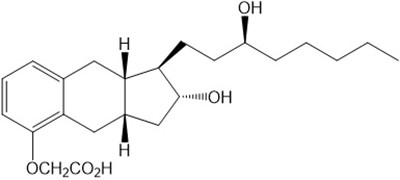
Product Images Tyvaso Dpi
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 65 images provide visual information about the product associated with Tyvaso Dpi NDC 66302-630 by United Therapeutics Corporation, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - tyvasoDPI 02
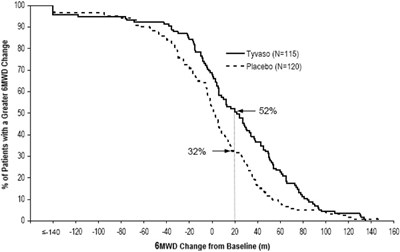
The text describes a comparison of the percentage of patients with a greater MWD (maximum walking distance) change between a drug called Tyvaso (with a sample size of 115) and a placebo (with a sample size of 120). The comparison is shown through a graph with a y-axis scale of <140, 120, 80, and 40, and an x-axis with increments of 20 from 0 to 100. The chart shows that the MWD change from baseline (in meters) is higher for the Tyvaso group compared to the placebo group.*
Figure 2 - tyvasoDPI 03

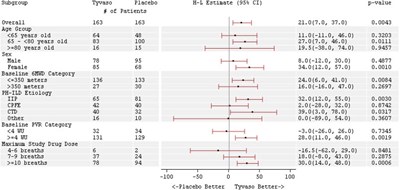
This text presents the results of a clinical trial that compared the effect of a medication called Tyvaso versus a placebo in individuals with pulmonary arterial hypertension. The trial included a total of 120 subjects, with different ages and gender, but mainly from North America. The results show several variables, such as Baseline 6MWD (distance walked in six minutes), different age quartiles, etiology, and background therapy. Additionally, the table presents the Placebo-Corrected Median Treatment Effect of Tyvaso.*
Figure 3 - tyvasoDPI 04

This appears to be a table showing study visit timelines and results of a medication's effects in weeks 4, 12, 15, and 16 on subjects. It also includes confidence interval percentages and median treatment effects in a placebo-controlled setting.*
Figure D - tyvasoDPI 10
This appears to be a list of doses, measured in mcg (micrograms), possibly for a medication or supplement. However, without additional context or information, it is not clear what these doses refer to or how they should be used.*
Image - tyvasoDPI 19

The text is not available as it only includes color and a single word that is difficult to determine the context.*
Figure I - tyvasoDPI 23

The text provides information about two printer cartridges, one dark blue and one light blue. Additionally, it indicates that the memory or capacity of the cartridges is 32 megabytes and 48 megabytes respectively. There is insufficient information to determine what type of cartridges they are, what brand they belong to, or what printer they are compatible with.*
PRINCIPAL DISPLAY PANEL - 16 mcg Institutional Kit - tyvasoDPI 55

TYVASO DPI is a medication containing treprostinil used for oral inhalation. It is available in an institutional kit, which includes multiple cartridges. It is not suitable for everyone and should only be used as prescribed by a medical professional. Some characters are not readable in the given text.*
PRINCIPAL DISPLAY PANEL - 32 mcg Institutional Kit - tyvasoDPI 57

This is a description for the Institutional Kit of TYVASO DPI, which is used for oral inhalation only. The kit contains 16 cartridges, each with 32 meg per cartridge. It is not clear what the active ingredient is due to errors.*
PRINCIPAL DISPLAY PANEL - 48 mcg Institutional Kit - tyvasoDPI 59

The text is a description of an "Institutional Kit" for oral inhalation. The kit includes 16 cartridges, each containing 48 meg per cartridge. The kit is not recommended for ventilation. The text also mentions "TYVASO DPI Sprepay | ks" (reprostni). The text provides some details related to usage and replacement of the inhalers.*
PRINCIPAL DISPLAY PANEL - 64 mcg Institutional Kit - tyvasoDPI 61

This appears to be a description of an Institutional Kit intended for oral inhalation only. The kit contains 16 cartridges, each containing 84 meg per cartridge inhalers of TYVASO DPI (treprostinil) recommended dosage of 820 Pressing information. The instructions on how to use the kit are also included.*
PRINCIPAL DISPLAY PANEL - 16 mcg 32 mcg 48 mcg Titration Kit - tyvasoDPI 65

The text seems to be a product label for a Titration Kit called "TYVASO DPI". It may contain a drug ingredient called "treprostini" and some identification codes. However, some characters are not readable, so complete information cannot be obtained.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.