Product Images Hydrocodone Bitartrate And Acetaminophen
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Hydrocodone Bitartrate And Acetaminophen NDC 66336-408 by Dispensing Solutions, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
NDC 66336-0408-XX - NDC 66336 0408 XX Qualitest

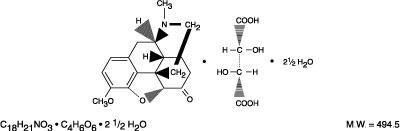
This is a description of a bulk source data for Qualitest Pharmaceuticals regarding a pink capsule-shaped tablet with debossed 3600 /V. The bulk source NDC is 00603-3888-28, with MFR lot XXXXXX and pedigree#858300. The tablet should be dispensed in a tight/light-resistant container, taken orally every few hours as needed for pain, and taken with food, but not with alcohol. This medication may cause drowsiness and impair the ability to drive. Each tablet contains hydrocodone bitartrate USP 10 mg and acetaminophen USP 500 mg. It may be habit-forming, and CAUTION: federal law prohibits the transfer of this drug to any person other than the patient for whom it was prescribed. The tablets should be kept out of children's reach and stored at 68°-77°F. This medication comes in C3 tablets only with an expiration date of 00-00. Packaged exclusively by Dispensing Solutions, Santa Ana, CA 92704.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.