FDA Label for Emvita 8
View Indications, Usage & Precautions
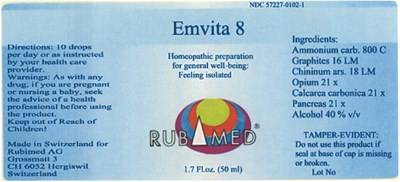
Emvita 8 Product Label
The following document was submitted to the FDA by the labeler of this product Rubimed Ag. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Otc - Purpose
Homeopathic preparation for general well-being: Feeling isolated
Inactive Ingredient
Ammonium carb. 800 C
Graphites 166 IA
Chininum ars. 18 LM
Opium 21 x
Calcarea carbonica 21 x
Pancreas 21 x
Alcohol 40 % v/v
Tamper-Evident
Do not use this product if seal at base of cap is missing or broken.
Directions
10 drops per day or as instructed by your health care provider.
Otc - Pregnancy Or Breast Feeding
As with any drug, if you are pregnant or nursing a baby, seek the advice of a health professional before using the product.
Otc - Keep Out Of Reach Of Children
Keep out of Reach of Children!
Principal Display Panel - 50 Ml Bottle Label
Emvita 8
Homeopathic preparation
for general well-being:
Feeling isolated
RUBIMED®
1.7 Fl.oz. (50 ml)
* Please review the disclaimer below.