FDA Label for Instant Cool Skin Instant Relief. Sudden Results
View Indications, Usage & Precautions
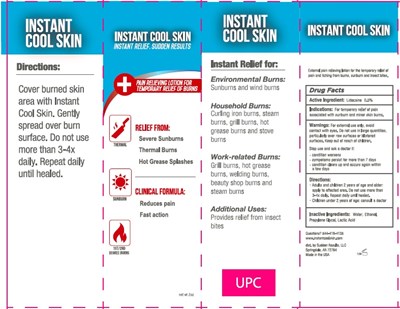
Instant Cool Skin Instant Relief. Sudden Results Product Label
The following document was submitted to the FDA by the labeler of this product Therapon Skin Health, Lp. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredients
Lidocaine 0.5%
Uses
External Analgesic
Indications
For temporary relief of pain associated with sunburn and minor skin burns.
Warnings
For external use only, avoid contact with eyes. Do not use in large quantities, particularly over raw surfaces or blistered surfaces
Directions
- Adults and children 2 years and older: apply to affected area. Do not use more than 3-4x daily. Repeat daily until healed.
- Children under 2 years of age: consult a doctor
Inactive Ingredients
Water, Ethanol, Propylene Glycol, Lactic Acid
Package Display
* Please review the disclaimer below.