Product Images Sevoflurane
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Sevoflurane NDC 66794-015 by Piramal Critical Care Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
label100ml - sev 100ml

Sevoflurane, USP is an inhalation anesthetic contained in a 100mL bottle with usual dosage information available in package insert. It is manufactured in the USA by Piramal Critical Care, Inc. and should be stored between 20° to 25°C (68° to 77°F) with excursions permitted to 15° to 30" (59° to 86°F). The NDC is 66794-015-10 with lot number YON 1017.*
figure1 - sevo fig1
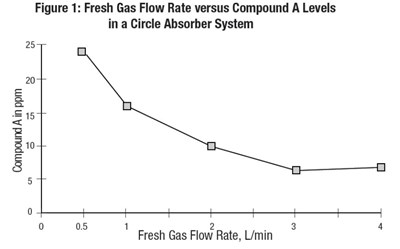
This is a graph displaying the Fresh Gas Flow Rate versus the levels of Compound A in parts per million (ppm) in a Circle Absorber System. The graph includes an x-axis labeled "Fresh Gas Flow Rate, L/min" and a y-axis labeled "Compound A in ppm". There is also a key indicating that the line on the graph corresponds to Compound A levels. The remaining text appears to be non-English characters or errors.*
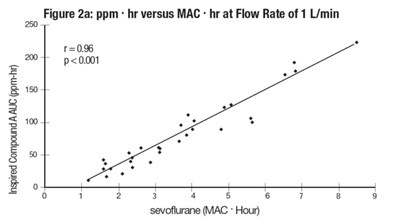
figure2 - sevo fig2
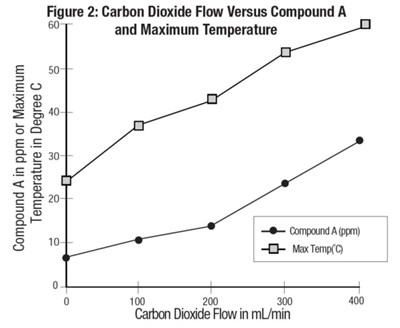
The image shows a graph illustrating the relationship between Carbon Dioxide flow and Compound A in parts per million (ppm), given at maximum temperature in degrees Celsius. The X-axis shows the Carbon Dioxide flow rate in milliliters per minute, while the two Y-axes show Compound A in ppm and Maximum Temperature in degrees Celsius, respectively. The graph seems to have been generated via an experimental process aimed at understanding the effect of Carbon Dioxide flow rate on Compound A concentrations at different temperatures.*
sevo fig3
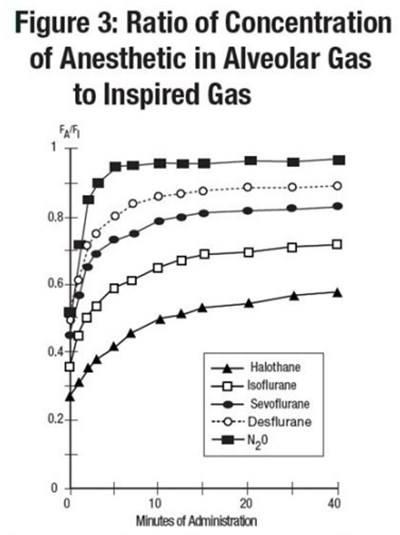
The text shows a figure labeled "Figure 3" displaying the concentration ratio of various anesthetics in alveolar gas to inspired gas. The anesthetics displayed are Halothane, Isoflurane, Sowfurane, and Desflurane. The values for the concentration ratio range from 0.08 to 0.04.*
sevo fig4

The image shows the concentration of different anesthetics in alveolar gas after the anesthesia has ended. The four anesthetics depicted are:FyFay, Halothane, Boflurane, Sewflurane, and Desflurane.*
sevo-fig-5 - sevo fig5
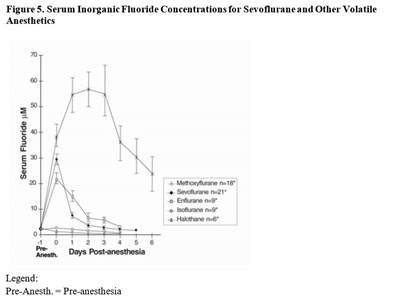
The figure shows the serum inorganic fluoride concentrations for sevoflurane and other volatile anesthetics. The x-axis represents the days post-anesthesia while the y-axis represents the serum fluoride concentration in micromoles per liter (uM). The legend indicates that "Pre-Anesth." refers to the serum fluoride concentration before anesthesia.*
sevo-fig-6 - sevo fig6

This is a figure showing the mean fluoride ion concentrations following the administration of sevoflurane at a mean minimum alveolar concentration (MAC) of 1.27 and mean duration of 2.06 hours. The plot includes fluoride ion concentration measurements at different time points represented in hours. The concentration measurements are shown on the y-axis in units of M (Moles per liter) while time is represented on the x-axis. There were 48 measurements used to calculate the mean fluoride ion concentration at each time point.*
sevo-fig-7 - sevo fig7

This is a graph showing the heart rate in beats per minute. The heart rate is plotted on the y-axis and ranges from 0 to 120 beats per minute. The x-axis has two labels, "A-Sevol0" and "A C-Sevo/Oa/Ny0". The heart rate peaks at around 120 beats per minute and decreases steadily until it reaches around 80 beats per minute. There is also a reference to a "BASELINE MAC LEVEL 1 15 2", which may refer to a measurement or setting related to the heart rate.*
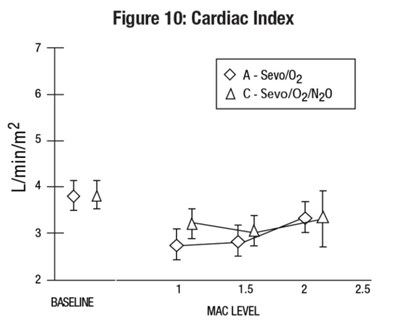
sevo-fig-8 - sevo fig8

The text does not provide enough information to generate a useful description as it consists of only some numbers, special characters, and abbreviations. It appears to be a graph showing Mean Arterial Pressure with a baseline and MAC level, but the information is incomplete.*
sevoflurane 250ml

This is a description of a medication called Sevoflurane. It is an inhalation anesthetic that is used for inhalation anesthesia. The packaging contains 250 mL of the medication and usual dosage instructions can be found on the package insert. The medication should be stored within a certain temperature range. Piramal Critical Care, Inc manufactures it. No further details are available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.