Product Images Gablofen
View Photos of Packaging, Labels & Appearance
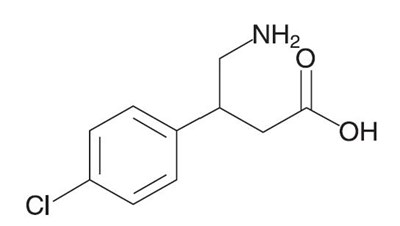
- structure - gablofen structure
- syringecrt2000mcg - gablofen syringe carton 2000mcg
- syringcrt500mcg - gablofen syringe carton 500mcg
- syringecrt50mcg - gablofen syringe carton 50mcg
- syringelbl1000mcg - gablofen syringe label 1000mcg
- syringelbl2000mcg - gablofen syringe label 2000mcg
- syringelbl500mcg - gablofen syringe label 500mcg
- syringelbl50mcg - gablofen syringe label 50mcg
- gablofen syringecarton 1000mcg
- tray1000mcg - gablofen tray 1000mcg
- tray2000mcg - gablofen tray 2000mcg
- tray500mcg - gablofen tray 500mcg
- tray50mcg - gablofen tray 50mcg
- vialcrt1000mcg - gablofen vial carton 1000mcg
- vialcrt2000mcg - gablofen vial carton 2000mcg
- vialcrt500mcg - gablofen vial carton 500mcg
- viallbl1000mcg - gablofen vial label 1000mcg
- viallbl2000mcg - gablofen vial label 2000mcg
- viallbl500mcg - gablofen vial label 500mcg
Product Label Images
The following 19 images provide visual information about the product associated with Gablofen NDC 66794-155 by Piramal Critical Care Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
syringecrt2000mcg - gablofen syringe carton 2000mcg
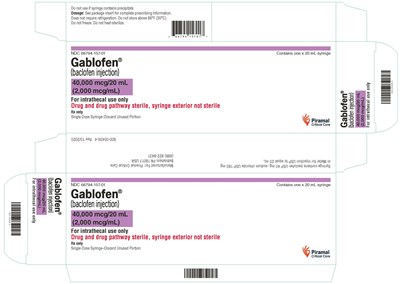
This is a drug called Gablofen, an injection of baclofen for intrathecal use only. It comes in a single-use, 20 mL syringe. The drug and drug pathway are sterile, but the syringe's exterior is not sterile. The dosage information is available in the package insert. The drug does not require refrigeration, but it should not be stored above 86°F (30°C). It should not be frozen or heat sterilized. If the syringe contains precipitate, it should not be used.*
syringcrt500mcg - gablofen syringe carton 500mcg

This is a medication called Gablofen, which is an injectable form of baclofen for intrathecal use only. The package includes a single 20 mL syringe containing 10,000 mcg or 500 mcg per mL of baclofen. It is important not to use it if there is any precipitate in the syringe. The package insert should be consulted for complete prescribing information. It does not require refrigeration but should not be stored above 86°F (30°C) and should not be frozen or heat-sterilized. The drug and pathway are sterile, but the exterior of the syringe is not. Any unused portion should be discarded. The product is distributed by Piramal Critical Care and has an NDC code of 66794-155-01.*
syringecrt50mcg - gablofen syringe carton 50mcg
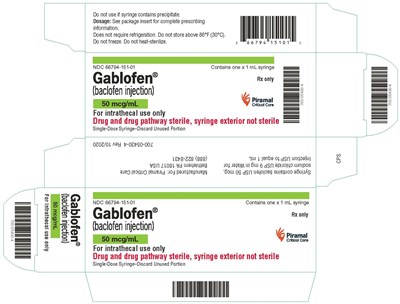
This is a description for Gablofen Ry, a medication which comes in a 1mL syringe and contains 50meg/mL Higamal, meant for intrathecal use only. It should not be used if the syringe contains precipitate, stored above 86°F (30°C), frozen, or heat-sterilized. The drug and drug pathway are sterile but the syringe exterior is not sterile. Unused portions in the syringe should be discarded. Complete prescribing information can be found in the package insert.*
syringelbl1000mcg - gablofen syringe label 1000mcg

Gablofen® is an injection containing baclofen in a sterile solution of 20,000 mcg/20 mL (1000 mcg/mL) intended for intrathecal use only. It is provided in a 20 mL syringe for maintenance dosing with an intrathecal pump. Any unused portion should be discarded, and it is only available on prescription. Piramal Critical Care manufactures it for the US from Bethlehem, PA.*
syringelbl2000mcg - gablofen syringe label 2000mcg
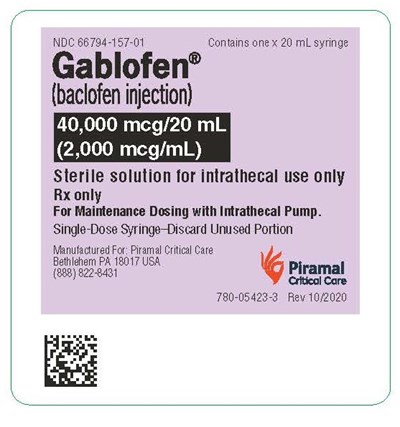
Gablofen® is a sterile solution of baclofen injection used for intrathecal purposes only. The package contains a single-dose syringe of 20 mL with 40,000 mcg/20 mL (2,000 meg/m) strength. It is intended for maintenance dosing with intrathecal pumps and the unused content should be discarded after single-use. This product is available only with a prescription and has been manufactured by Piramal Critical Care in Bethlehem, PA, 18017 USA. For any queries related to this product, one can contact Piramal at (888) 8228431.*
syringelbl500mcg - gablofen syringe label 500mcg
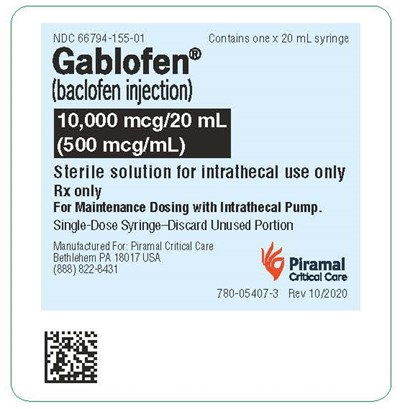
Gablofen is a sterile solution of baclofen injection, provided in a single-dose syringe of 20 mL, containing 10,000 meg/20mL or 500mcg/m. It is indicated for the maintenance dosing with an intrathecal pump and is for intrathecal use only, available via prescription only. Any unused portion of the syringe must be discarded. This medication is manufactured for Piramal Critical Care, located at Bethlehem PA 18017 USA.*
syringelbl50mcg - gablofen syringe label 50mcg
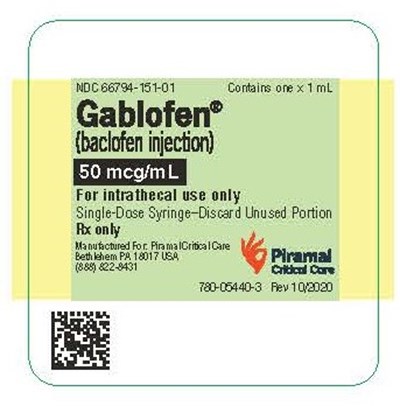
Gablofen® is an injection drug containing baclofen, which is used only for intrathecal use. Its presentation is in the form of single-dose syringes, and any unused portion should be discarded. The text also includes some alphanumeric codes that may relate to the product's lot number or other manufacturing details.*
tray1000mcg - gablofen tray 1000mcg

This is a product description for Gablofen, a sterile solution containing Baclofen for intrathecal use, sold in a 20 mL syringe. The drug comes in a concentration of 1,000 mcg/mL, with 20,000 mcg of Baclofen in each syringe. This solution is only for intrathecal use and not for injection into veins or muscles. The syringe's exterior is not sterile, and the product is packaged in Bethlehem, PA. Gablofen is a prescription medication used for maintenance dosing with intrathecal pumps. Contact (888) 622-8431 for more information.*
tray2000mcg - gablofen tray 2000mcg

This is a description of Gablofen, the brand name of baclofen injection. The drug is a sterile solution intended for intrathecal use only and is used for maintenance dosing with intrathecal pump. The syringe is a single-dose type and should be discarded if any portion remains unused. The exterior of the syringe is not sterile, but the drug and drug pathway are sterile. The product is manufactured by Piramal Critical Care and is available in 20 mL syringes. This product is prescription-only. The text also includes the NDC number for identification purposes.*
tray500mcg - gablofen tray 500mcg

Gablofen® is a sterile solution for intrathecal use only, manufactured by Piramal Critical Care. It contains 10,000 mcg/20 mL (500 mcg/mL) of baclofen and comes in a single 20 mL syringe. The drug and pathway are sterile, but the syringe exterior is not sterile. It is intended for maintenance dosing with an intrathecal pump and is available by prescription only. Contact Piramal Critical Care for more information.*
tray50mcg - gablofen tray 50mcg
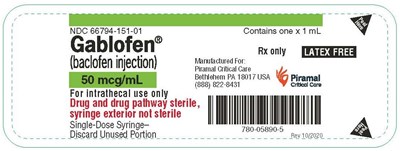
This is a description of a drug called Gablofen, which is an injection containing Baclofen. The injection is sold by Piramal Critical Care and comes in a pre-filled syringe. The syringe is not sterile on the outside and should be discarded after use. The NDC number is 66794-151-01 and each syringe contains 1mL of the drug.*
vialcrt1000mcg - gablofen vial carton 1000mcg
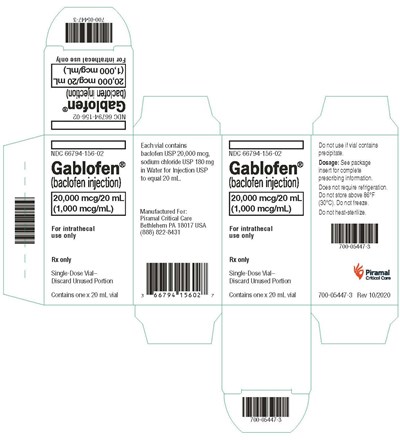
Gablofen® is a prescription medicine that comes in a single-dose vial of 20 mL for intrathecal use only. It contains baclofen USP 20,000 mg, sodium chloride USP 180 mg in Water for Injection USP, and is manufactured by Piramal Critical Care. The medication requires a prescription and should be discarded if unused after opening. The complete prescribing information is available in the package insert. Do not store the medication above 86°F (30°C) or freeze it, but it does not require refrigeration. The text also contains some identification numbers and a warning against using the medicine if the vial contains precipitate.*
vialcrt2000mcg - gablofen vial carton 2000mcg
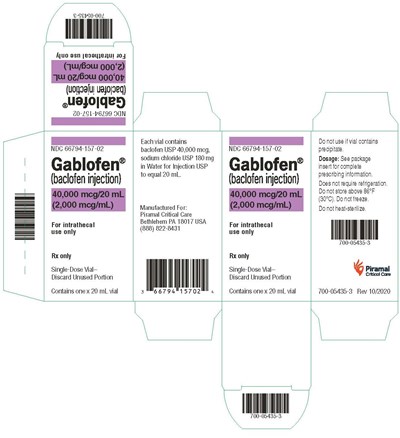
Gablofen is a prescription-only injectable medication for intrathecal use, meaning it is injected into the spinal cord. It is available in single-dose vials of 20 mL each, with instructions to discard any unused portion. Each vial contains 40,000 meg of baclofen, 180 mg of sodium chloride, and water for injection. It is important to not use the medication if the vial contains any precipitate. Dosage instructions are provided in the package insert. Gablofen should not be stored above 86°F and must not be frozen or heated for sterilization. The manufacturer for this medication is Piramal Critical Care, located in Bethlehem, PA.*
vialcrt500mcg - gablofen vial carton 500mcg
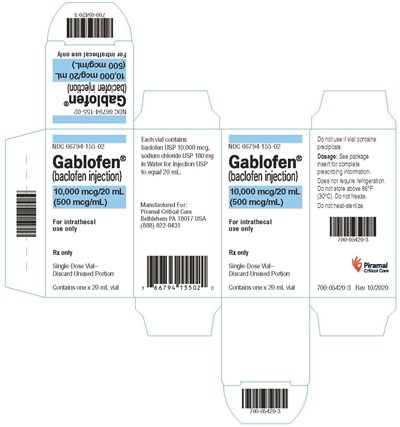
This is a pharmaceutical product description for Gablofen, which is an injectable form of baclofen. It is intended for intrathecal use only and comes in a single-dose vial. Each vial contains 10,000 mcg of baclofen USP, along with sodium chloride USP and water for injection USP, making up 20 mL. The manufacturer is Piramal Critical Care, based in Bethlehem, PA. The product should not be used if the vial contains precipitate and should not be heated for sterilization. Dosage instructions can be found in the package insert. The product does not require refrigeration but should not be stored above 86°F (30°C) or frozen.*
viallbl1000mcg - gablofen vial label 1000mcg

This is a description of medication called Gablofen®. It is an injection containing baclofen used for intrathecal use only. The text provides information on the dosage and single-dose vial instructions. The medication is manufactured by a company named Piramal.*
viallbl2000mcg - gablofen vial label 2000mcg

This is a description of a medication called Gablofen. It is a baclofen injection with a strength of 40,000 mcg/20 mL (2,000 mcg/mL). It is for intrathecal use only and requires a prescription. The medication comes in a single-dose vial with a discarded unused portion. The manufacturer is Piramal Critical Care. No additional information is available due to the poor quality of the .*
viallbl500mcg - gablofen vial label 500mcg

This is a description for an injection called Gablofen, specifically the 10,000 mcg/20 mL variant, manufactured by Piramal. The injection contains baclofen and is meant for critical care purposes. The packaging is a single-use vial, and any unused portion must be discarded. The rest of the text appears to be a product code and serial number.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.