Product Images Fluticasone Propionate Hfa
View Photos of Packaging, Labels & Appearance
- Fluticasone propionate chemical structure - fluticasonepropionatehfa spl graphic 01
- Figure 1. A 12-Week Clinical Trial in Subjects Aged 12 Years and Older Inadequately Controlled on Bronchodilators Alone: Mean Percent Change from Baseline in FEV1 Prior to AM Dose (Trial 1) - fluticasonepropionatehfa spl graphic 02
- Figure 2. A 12-Week Clinical Trial in Subjects Aged 12 Years and Older Already Receiving Daily Inhaled Corticosteroids: Mean Percent Change from Baseline in FEV1 Prior to AM Dose (Trial 2) - fluticasonepropionatehfa spl graphic 03
- Figure 3. A 16-Week Clinical Trial in Subjects Aged 12 Years and Older Requiring Chronic Oral Prednisone Therapy: Change in Maintenance Prednisone Dose - fluticasonepropionatehfa spl graphic 04
- Figure A - fluticasonepropionatehfa spl graphic 05
- Figure B - fluticasonepropionatehfa spl graphic 06
- Figure C - fluticasonepropionatehfa spl graphic 07
- Figure D - fluticasonepropionatehfa spl graphic 08
- Figure E - fluticasonepropionatehfa spl graphic 09
- Figure F - fluticasonepropionatehfa spl graphic 10
- Figure G - fluticasonepropionatehfa spl graphic 11
- Figure H - fluticasonepropionatehfa spl graphic 12
- Figure I - fluticasonepropionatehfa spl graphic 13
- Figure J - fluticasonepropionatehfa spl graphic 14
- Fluticasone Propionate HFA 44 mcg 120 dose carton - fluticasonepropionatehfa spl graphic 15
- Fluticasone Propionate 110 mcg 120 dose carton - fluticasonepropionatehfa spl graphic 16
- Fluticasone Propionate HFA 220 mcg dose carton - fluticasonepropionatehfa spl graphic 17
Product Label Images
The following 17 images provide visual information about the product associated with Fluticasone Propionate Hfa NDC 66993-078 by Prasco Laboratories, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. A 12-Week Clinical Trial in Subjects Aged 12 Years and Older Inadequately Controlled on Bronchodilators Alone: Mean Percent Change from Baseline in FEV1 Prior to AM Dose (Trial 1) - fluticasonepropionatehfa spl graphic 02
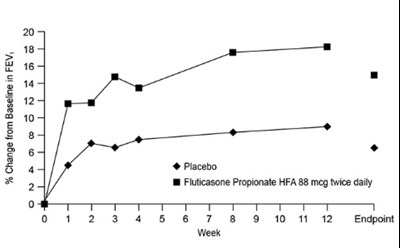
Figure 2. A 12-Week Clinical Trial in Subjects Aged 12 Years and Older Already Receiving Daily Inhaled Corticosteroids: Mean Percent Change from Baseline in FEV1 Prior to AM Dose (Trial 2) - fluticasonepropionatehfa spl graphic 03
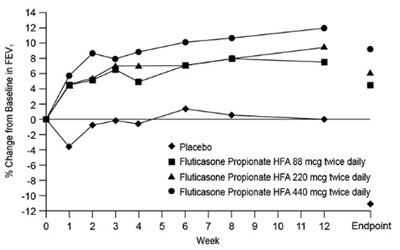
This appears to be a chart or graph related to a study on the effects of Fluticasone Propionate inhalers at different dosages on FEV (forced expiratory volume) in comparison to a placebo. The x-axis represents the weeks of the study, while the y-axis measures the percentage change in FEV from the baseline. Unfortunately, the chart is not readable due to the presence of special characters and typos obtained by the .*
Figure 3. A 16-Week Clinical Trial in Subjects Aged 12 Years and Older Requiring Chronic Oral Prednisone Therapy: Change in Maintenance Prednisone Dose - fluticasonepropionatehfa spl graphic 04
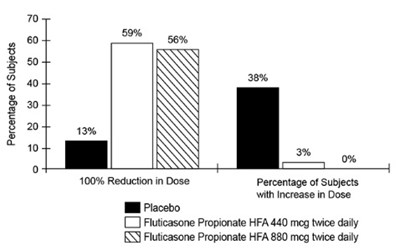
The text seems to be showing data related to the percentage of subjects with an increase in dose and a reduction in dose for Fluticasone Propionate HFA medication. The percentage of subjects with a reduction in dose is not specified but there are percentages given for different dose levels. The term "Il Piacebo" is not clear in this context and could be a typo.*
Figure A - fluticasonepropionatehfa spl graphic 05
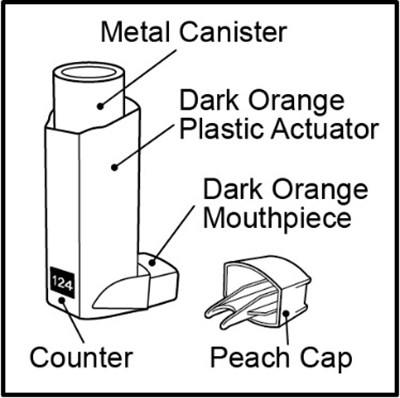
This is a description of a metal canister that comes with a dark orange plastic actuator and mouthpiece. The canister is closed off by a peach-colored cap.*
Figure E - fluticasonepropionatehfa spl graphic 09
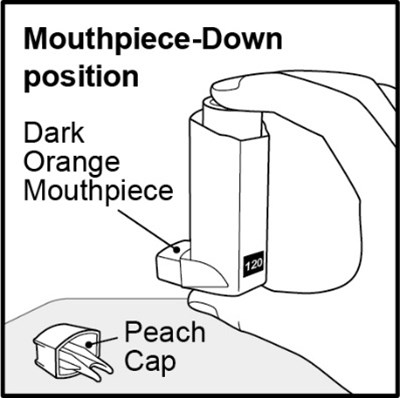
The text appears to be describing the position of a mouthpiece that is down, with a color appearing to be dark orange. It is not clear what item this mouthpiece belongs to, but it could potentially be a musical instrument or a breathing device.*
Figure F - fluticasonepropionatehfa spl graphic 10
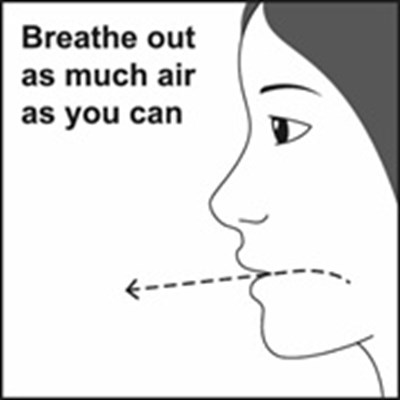
This text seems to be a simple instruction to breathe out as much air as one can.*
Fluticasone Propionate HFA 44 mcg 120 dose carton - fluticasonepropionatehfa spl graphic 15

This is a description of a medication called Fluticasone Propionate HFA Inhalation Aerosol, manufactured by Prasco, with NDC code 66993-076-96. It is an inhaler that contains a suspension of micronized fluticasone propionate in a propellant called HFA-134a. Each actuation of the inhaler delivers 50 mcg of fluticasone propionate from the valve and 44 mcg from the actuator. The net weight is 10.6g, and it offers 120 metered actuations. It is important to note that the inhaler should only be used with the Fluticasone Propionate HFA actuator.*
Fluticasone Propionate 110 mcg 120 dose carton - fluticasonepropionatehfa spl graphic 16

This is a medication with the National Drug Code (NDC) of 66993-079-96 manufactured by PRASCO. The medication is an inhalation aerosol of Fluticasone Propionate HFA which is only intended for oral inhalation with a Fluticasone Propionate HFA actuator. The canister contains a microcrystalline suspension of fluticasone propionate in propellant HFA-134a. Each actuation of the inhaler delivers 125 mcg of fluticasone propionate from the valve and 110 mcg from the actuator. The medication has a net weight of 12g and can deliver 120 metered actuations.*
Fluticasone Propionate HFA 220 mcg dose carton - fluticasonepropionatehfa spl graphic 17

This is a description of a medication called Fluticasone Propionate HIFA, which is an inhalation aerosol used for oral inhalation with a specific Fluticasone Propionate HFA actuator. It contains a microcrystalline suspension of micronized fluticasone propionate in propellant HFA-134a, and each actuation delivers 250 mcg of fluticasone propionate from the valve and 220 mcg from the actuator. The medication is available in a canister with a net weight of 12 g and 120 metered actuations.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.







