Product Images Fluticasone Propionate Diskus
View Photos of Packaging, Labels & Appearance
- Fluticasone propionate chemical structure - fluticasone propionate diskus spl graphic 01
- Figure 1 - fluticasone propionate diskus spl graphic 02
- Figure 2 - fluticasone propionate diskus spl graphic 03
- Figure 3 - fluticasone propionate diskus spl graphic 04
- Figure 4 - fluticasone propionate diskus spl graphic 05
- Figure A - fluticasone propionate diskus spl graphic 06
- Figure B - fluticasone propionate diskus spl graphic 07
- Figure C - fluticasone propionate diskus spl graphic 08
- Figure D - fluticasone propionate diskus spl graphic 09
- Figure E - fluticasone propionate diskus spl graphic 10
- Figure F - fluticasone propionate diskus spl graphic 11
- Figure G - fluticasone propionate diskus spl graphic 12
- Figure H - fluticasone propionate diskus spl graphic 13
- Fluticasone Propionate Diskus 50 mcg 60 dose carton - fluticasone propionate diskus spl graphic 14
- Fluticasone Propionate Diskus 100 mcg 60 dose carton - fluticasone propionate diskus spl graphic 15
- Fluticasone Propionate Diskus 250 mcg 60 dose carton - fluticasone propionate diskus spl graphic 16
Product Label Images
The following 16 images provide visual information about the product associated with Fluticasone Propionate Diskus NDC 66993-792 by Prasco Laboratories, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - fluticasone propionate diskus spl graphic 02
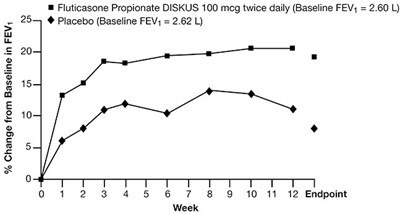
This text appears to be a description of a study or experiment involving the use of Fluticasone Propionate DISKUS at a dosage of 100 mcg twice daily. The baseline forced expiratory volume (FEV) for the participants was 2.60 L. The text also mentions a placebo group with a baseline FEV of 2.62 L. The " % Change from Baseline in FEV" is mentioned as well. The text includes a chart or timeline with various timepoints labeled from 0 to 12. However, without additional context or information, it is difficult to provide a more detailed description of the content.*
Figure 2 - fluticasone propionate diskus spl graphic 03
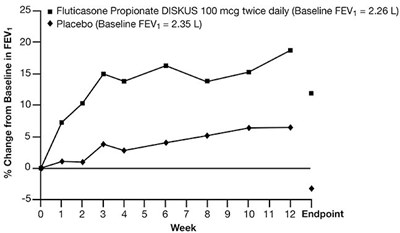
The text appears to be a description of a medical evaluation involving the use of Fluticasone Propionate DISKUS at a dosage of 100 micrograms twice daily. The evaluation includes measuring FEV (Forced Expiratory Volume) at baseline, with a starting value of 2.26 L. There is also a mention of a placebo with a baseline FEV value of 2.35. The text indicates a percentage change from baseline but does not provide specific information about the change. The evaluation seems to have a time frame of 12 weeks, as indicated by the endpoints mentioned at 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, and 12 weeks.*
Fluticasone Propionate Diskus 50 mcg 60 dose carton - fluticasone propionate diskus spl graphic 14
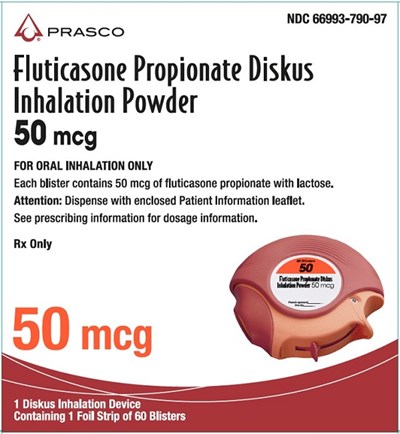
This text appears to be a description of a medication called Fluticasone Propionate Diskus, which is an inhalation powder used for oral inhalation. Each blister contains 50 micrograms of fluticasone propionate with lactose. The package includes 1 Diskus Inhalation Device containing 1 foil strip of 60 blisters. The text also mentions that the patient should refer to the enclosed Patient Information leaflet for instructions and dosage information.*
Fluticasone Propionate Diskus 100 mcg 60 dose carton - fluticasone propionate diskus spl graphic 15

This is a description of a medication called Fluticasone Propionate Diskus Inhalation Powder. It is intended for oral inhalation only and each blister contains 100 mcg of fluticasone propionate with lactose. It is dispensed with an enclosed Patient Information leaflet and the dosage information can be found in the prescribing information. The package includes 1 Diskus Inhalation Device containing 1 Foil Strip of 60 Blisters.*
Fluticasone Propionate Diskus 250 mcg 60 dose carton - fluticasone propionate diskus spl graphic 16

This text is a product description for a medication called "Fluticasone Propionate Diskus". It is an inhalation powder used for oral inhalation only. Each blister contains 250 mcg of fluticasone propionate with lactose. The product comes with a Diskus inhalation device that contains 1 foil strip of 60 blisters. The text also mentions an enclosed Patient Information leaflet and advises to refer to the prescribing information for dosage details.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.










