Product Images Gabapentin
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Gabapentin NDC 67046-341 by Contract Pharmacy Services-pa, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
g6 - Gabapentin 600mg 341

This is a description of a medication called Gabapentin available as 600 mg tablets in a bottle of 30. It has a lot number, pill identification number, and an expiry date of December 31, 2020. It is manufactured by Acihealthcare and has a SIN and batch number. Dosage instructions can be found in the package insert and it is to be stored at a controlled room temperature between 20-25°C (68-77°F) with permitted excursions to 15-30°C (50-86°F). The medication is packaged by Cantract Pharmacy Servines-PA and has an NDC of 67046-341-30.*
g8 - Gabapentin 800mg 413

Gabapentin is a prescription-only medication available in tablet form. Each tablet contains 800mg of Gabapentin, and each package contains 30 tablets. The expiration date of the medication is 12/31/2020. This medication is manufactured in accordance with USP standards. The usual dose is only available through the full prescribing information. The tablets can be stored at a temperature of 20-25°C (68-77°F), but excursions to 15-30°C (59-86°F) are permitted. The packaging was done by Contract Pharmacy Services-PA. The National Drug Code (NDC) for the 30-tablet package is 67046-413-30. The Lot and Batch numbers have been masked for privacy.*
Figure 1 - ab1e0185 b802 4cf2 a5d4 b71f27f1dcb0 00
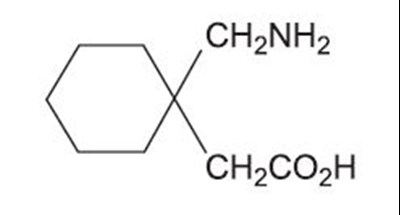
This text describes a formula for calculating the estimated creatinine clearance (CLOr) of a patient. It involves multiplying the patient's age (in years) by their weight (in kg) and dividing by a constant value of 72 multiplied by their serum creatinine level (in mg/dL). For female patients, an additional step of multiplying by 0.85 is required.*
figure02 - d45f499b 8612 479e 8dd2 4fe28b1f5f9c 03
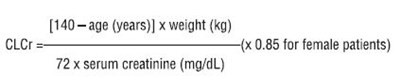
This text is providing the results of a study evaluating pain scores over a 4-week fixed dose period for two different treatments, Gabapentin at 1800 mg/day and Gabapentin at 2400 mg/day, compared to a placebo. The data includes mean pain scores at baseline and throughout the 7-day evaluation period.*
figure03 - d45f499b 8612 479e 8dd2 4fe28b1f5f9c 04
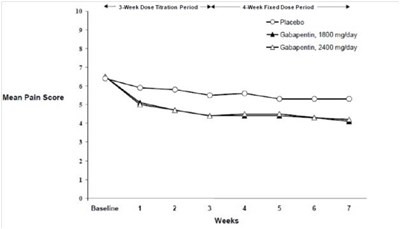
This is a chart showing the proportion of responders who experienced more than 50% reduction in pain score in controlled PHN studies. There are two studies presented and both have a significant p-value of less than 0.001. The data presents the percentage of responders for each study, one of which had 32% responders while the other had 34%. The chart also includes information on the cost of treatment with GBP 3600, GBP 1800, and GBP 2400.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.