Product Images Plavix
View Photos of Packaging, Labels & Appearance
- Chemical Structure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 01
- Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 02
- Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 03
- Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 04
- Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 05
- Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 06
- Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 07
- Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 08
- Blister of 30 Tablets - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 09
Product Label Images
The following 9 images provide visual information about the product associated with Plavix NDC 67046-604 by Contract Pharmacy Services-pa, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 02
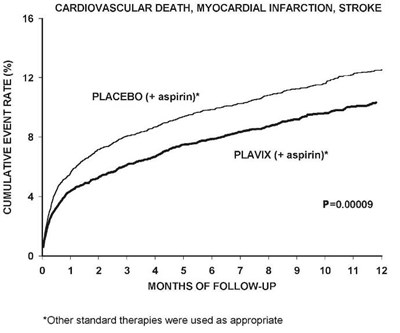
The text describes a chart that shows the cumulative event rate for cardiovascular death, myocardial infarction, and stroke for patients who were given either a placebo with aspirin or Plavix with aspirin over a 12-month follow-up period. The chart shows a significant difference between the two treatments, with Plavix having a lower cumulative event rate. Other standard therapies were also used.*
Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 03
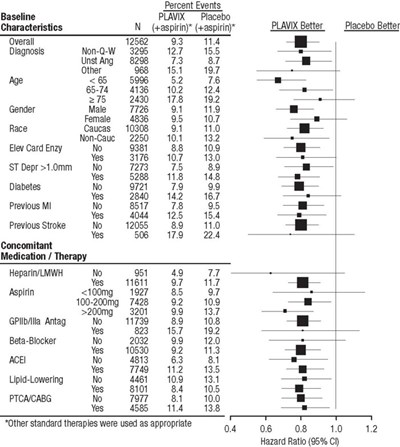
This appears to be a table of data showing different characteristics of a study group that took either Plavix or placebo. The characteristics include demographic information such as age, gender, and race as well as medical history such as previous heart attacks or strokes and concomitant medications or therapies. There is also mention of hazard ratios with corresponding confidence intervals.*
Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 04
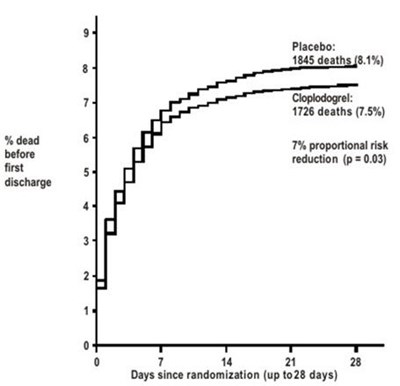
The text seems to be a comparison of the number of deaths before the first discharge in a placebo group versus a group that received Clopidogrel. The Clopidogrel group had a 7% proportional risk reduction (p=0.03) in deaths compared to the placebo group. The data is plotted over a span of up to 28 days since randomization.*
Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 05
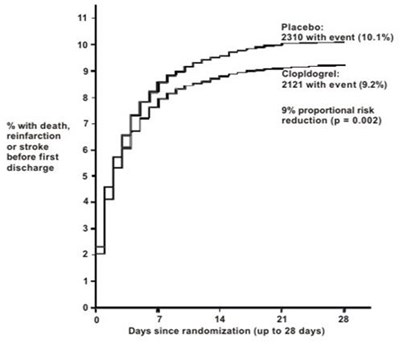
This text describes the results of a study comparing the effectiveness of a placebo versus the drug Tiopldogrel in reducing the risk of discharge for patients over a 28-day period. The results indicate that the proportion of patients with an event of discharge was 10.1% in the placebo group and 9.2% in the Tiopldogrel group, which represents a 9% proportional risk reduction with the drug (p=0.002). The data is presented graphically with the number of days since randomization.*
Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 06
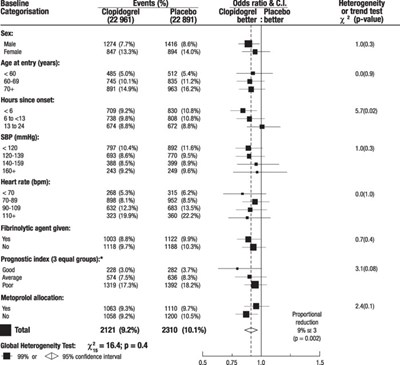
This appears to be a statistical analysis report of a clinical trial investigating the effectiveness of Clopidogrel Placebo in treating a medical condition. The report presents data on various subgroups of the trial population, including sex, age, blood pressure, and heart rate, as well as whether or not they were administered a fibrinolytic agent. The report also includes information on the Metoprolol allocation, with a reduction of 1% and a total percentage of 53%.*
Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 07
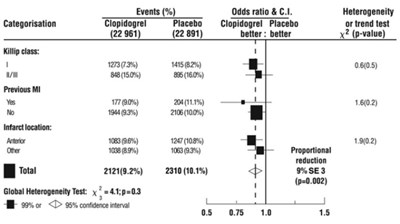
This is a table of results from a study that compared the use of Clopidogrel versus Placebo in patients with Killip class I and a previous myocardial infarction. The outcome measures include infarct location and global heterogeneity test. The results show that Clopidogrel was statistically significantly better than Placebo in reducing the proportional reduction of events (p=0.002) at a 95% confidence interval.*
Figure - e5a90643 3f0e 45a2 a9a4 65229c38d6ab 08
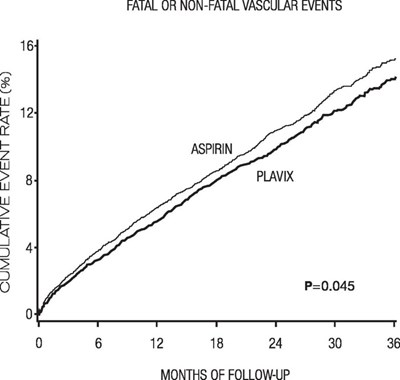
This text presents a graph showing the cumulative event rate in percentage for Faial or Non-Faial vascular events over a period of 36 months of follow-up. The graph compares the effects of two medications, Aspirin and Plavix.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

