Product Images Imatinib Mesylate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Imatinib Mesylate NDC 67184-0533 by Qilu Pharmaceutical Co., Ltd., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 4: Study 2 Recurrence-Free Survival (ITT Population) - imt 04

This appears to be a table summarizing some sort of medical or biological data, including measurements of survival time and recurrence rate. However, without additional context or information on the parameters being measured, it is difficult to provide a more specific description.*
Figure 5: Study 2 Overall Survival (ITT Population) - imt 05

The provided text includes information on a clinical trial, displaying a graph showing the overall survival rate of patients who were given Imatinib treatment over time. The graph shows that there is a hazard ratio of 0.45 demonstrating a reduction in mortality risk, and also includes survival rates at different intervals after treatment. The number 100 is also present but appears to be unrelated to the clinical trial description.*
PRINCIPAL DISPLAY PANEL – BOTTLE LABEL – 100 MG TABLETS NDC 0078-0401-34 imatinib mesylate tablets® (imatinib mesylate) Tablets 100 mg Rx only Each tablet contains 100 mg of imatinib free base. 90 Tablets - imt 100mg 500s label

Imatinib Mesylate is a prescription medication manufactured by Qilu Pharmaceutical (Hainan) Co. Ltd that contains 100 mg of imatinib free base per tablet. The dosage information can be found in the prescribing information. The tablets should be stored at a temperature between 20°C to 25°C (68°F to 77°F) and can be permitted to be exposed to temperatures between 15°C to 30°C (59°F to 86°F). It is important to protect the tablets from moisture and dispense them in a tight container. This medication is available in a container of 500 tablets and should be kept out of the reach of children. LOT and RX numbers are also provided but the patient-specific information is excluded.*
PRINCIPAL DISPLAY PANEL – BOTTLE LABEL – 100 MG TABLETS NDC 0078-0401-34 imatinib mesylate tablets® (imatinib mesylate) Tablets 100 mg Rx only Each tablet contains 100 mg of imatinib free base. 90 Tablets - imt 100mg 90s label

Each tablet contains 100 mg of imatinib free base manufactured by Qilu Pharmaceutical (Hainan) Co., Ltd. The dosage information can be found at the Prescribing Information. The tablets should be stored at temperatures between 20°C to 25°C (68°F to 77°F) to protect them from moisture. It is important to keep this and all drugs out of the reach of children. This medication is only available by prescription and has an NDC code of 67184-0532-1. The LOT number for this batch is X00KKKX and the TIN number is XXXKKKKXKKKNX.*
PRINCIPAL DISPLAY PANEL – CARTON LABEL – 100 MG TABLETS - imt 100mg carton

Imatinib Mesylate is a medication available in tablet form with 100mg dosage. The package is not child-resistant and intended for institutional use only. The package contains 30 tablets which are split into 3 cards each containing 10 tablets. The unit dose package is registered with the NDC 6718405323.*
PRINCIPAL DISPLAY PANEL – BOTTLE LABEL – 400 MG TABLETS - imt 400mg 300s label

Each tablet contains 400mg of imatinib free base manufactured by Qilu Pharmaceutical (Hainan) Co., Ltd. The recommended storage temperature for Imatinib Mesylate Tablets is 20°C to 25°C (68°F to 77°F) with excursions permitted to 15°C to 30°C (59°F to 86°F). Protect it from moisture and dispense in a tight container, USP. This medicine should be kept out of the reach of children. Dosage instructions can be found in the prescribing information. The text also includes the NDC number and the manufacturer's name, but a unique description cannot be created due to the lack of information.*
PRINCIPAL DISPLAY PANEL – BOTTLE LABEL – 400 MG TABLETS - imt 400mg 30s label

Imatinib mesylate tablets are available in a strength of 400 mg per tablet, manufactured by Qilu Pharmaceutical (Hainan) Co., Ltd in China. The prescribing information should be consulted for dosage instructions. The tablets should be stored at room temperature, protected from USP. This medication is for prescription use only, and must be kept out of reach of children. Each package contains 30 tablets.*
PRINCIPAL DISPLAY PANEL – CARTON LABEL – 400 MG TABLETS (imatinib mesylate) tablets 400 mg per tablet Each tablet contains 400 mg of imatinib free base 30 Tablets Rx only NOVARTIS - imt 400mg carton
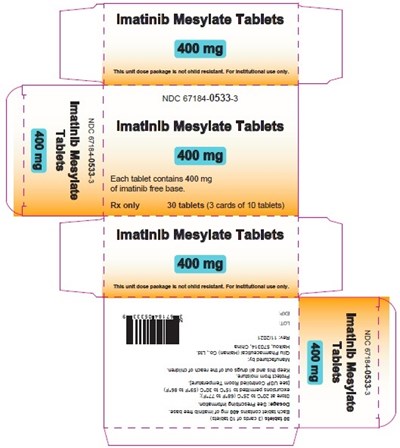
This is a description of Imatinib Mesylate Tablets, which come in 400mg doses. Each tablet contains 400mg of the free base of Imatinib. The packaging contains 30 tablets, packaged in 3 cards, each with 10 tablets. The rest of the text is not readable, due to errors.*
Figure 1 Progression Free Survival (ITT Principle) - imt figure01 free survival

This is a statistical analysis of patients with and without progression in a clinical trial that involved Imatinib and Ara-C as treatment options. The data shows the number of patients who experienced disease progression, patients who were censored at discontinuation, and patients who were censored at the last follow-up. The hazard ratio and log-rank test p-value are also provided. It appears that Imatinib Mesylate was the treatment arm being evaluated, but further details about the trial and its results are not available.*
Figure 2 Time to Progression to AP or BC (ITT Principle) - imt figure02 time to progression

This text presents data regarding the effectiveness of Imatinib Mesylate with or without 1FN+Ara-C in preventing progression to AP or BC. The table shows the number of patients who progressed to AP or BC, or were censored at discontinuation or last follow-up. The hazard ratio and log-rank test are also shown. The data is plotted against time since randomization.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.



