Product Images Haloperidol
View Photos of Packaging, Labels & Appearance
- Structural Formula - haloperidol tablets usp 01
- PRINCIPAL DISPLAY PANELNDC 58657-700-01Haloperidol Tablets, USP0.5 mgRx Only100 Tablets - haloperidol tablets usp 02
- PRINCIPAL DISPLAY PANELNDC 58657-700-10Haloperidol Tablets, USP0.5 mgRx Only1000 Tablets - haloperidol tablets usp 03
- PRINCIPAL DISPLAY PANELNDC 58657-701-01Haloperidol Tablets, USP1 mgRx Only100 Tablets - haloperidol tablets usp 04
- PRINCIPAL DISPLAY PANELNDC 58657-701-10Haloperidol Tablets, USP1 mgRx Only1000 Tablets - haloperidol tablets usp 05
- PRINCIPAL DISPLAY PANELNDC 58657-702-01Haloperidol Tablets, USP2 mgRx Only100 Tablets - haloperidol tablets usp 06
- PRINCIPAL DISPLAY PANELNDC 58657-702-10Haloperidol Tablets, USP2 mgRx Only1000 Tablets - haloperidol tablets usp 07
- PRINCIPAL DISPLAY PANELNDC 58657-703-01Haloperidol Tablets, USP5 mgRx Only100 Tablets - haloperidol tablets usp 08
- PRINCIPAL DISPLAY PANELNDC 58657-703-10Haloperidol Tablets, USP5 mgRx Only1000 Tablets - haloperidol tablets usp 09
- PRINCIPAL DISPLAY PANELNDC 58657-704-01Haloperidol Tablets, USP10 mgRx Only100 Tablets - haloperidol tablets usp 0a
- PRINCIPAL DISPLAY PANELNDC 58657-704-10Haloperidol Tablets, USP10 mgRx Only1000 Tablets - haloperidol tablets usp 0b
- PRINCIPAL DISPLAY PANELNDC 58657-705-01Haloperidol Tablets, USP20 mgRx Only100 Tablets - haloperidol tablets usp 0c
- PRINCIPAL DISPLAY PANELNDC 58657-705-10Haloperidol Tablets, USP20 mgRx Only1000 Tablets - haloperidol tablets usp 0d
Product Label Images
The following 13 images provide visual information about the product associated with Haloperidol NDC 67296-1462 by Redpharm Drug, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANELNDC 58657-700-01Haloperidol Tablets, USP0.5 mgRx Only100 Tablets - haloperidol tablets usp 02
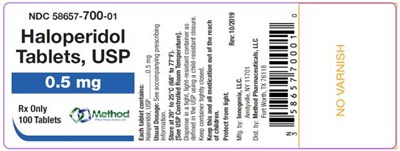
This is a medication with the National Drug Code (NDC) 58657-700-01. It is Haloperidol, in the form of tablets, and meets the United States Pharmacopeia (USP) standards. There are 100 tablets in each package. The significance of "BOW e\ cthod" is not clear.*
PRINCIPAL DISPLAY PANELNDC 58657-700-10Haloperidol Tablets, USP0.5 mgRx Only1000 Tablets - haloperidol tablets usp 03

This description is for a box of Haloperidol Tablets, USP which contains 1000 tablets. The NDC code is 58657-700-10 and the manufacturer's batch number is unclear as it appears to read "Bx nky Method". The text also includes information about the strength of the tablets, which is not discernible from the output. There is unclear text that mentions "Rewtong" and "e" which are not informative. The text appears to contain errors and is not comprehensive enough to provide a useful description.*
PRINCIPAL DISPLAY PANELNDC 58657-701-01Haloperidol Tablets, USP1 mgRx Only100 Tablets - haloperidol tablets usp 04
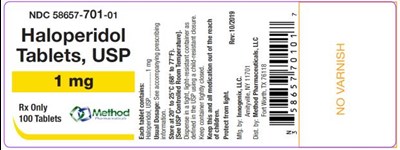
NDC 58657-701-01 is a prescription drug called Haloperidol. It is available in the form of tablets with a strength of 1 mg. The package contains 100 tablets and it can be obtained only on a prescription.*
PRINCIPAL DISPLAY PANELNDC 58657-701-10Haloperidol Tablets, USP1 mgRx Only1000 Tablets - haloperidol tablets usp 05
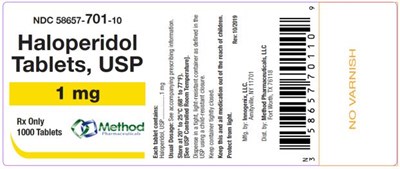
This is a description of a medication with NDC code 58657-701-10. The medication is Haloperidol that comes in the form of tablets with a strength of 1mg. The medication is only available with a prescription and it comes in a bottle that contains 1000 tablets.*
PRINCIPAL DISPLAY PANELNDC 58657-702-01Haloperidol Tablets, USP2 mgRx Only100 Tablets - haloperidol tablets usp 06
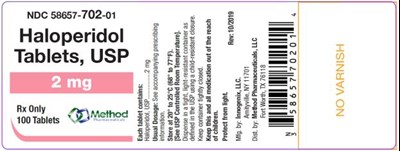
This is a description of Haloperidol Tablets, USP with NDC 58657-702-01 used only on prescription. The tablets come in a bottle containing 100 tablets and were revised on October 7, 2010. The manufacturer is Innogeni, LLC.*
PRINCIPAL DISPLAY PANELNDC 58657-702-10Haloperidol Tablets, USP2 mgRx Only1000 Tablets - haloperidol tablets usp 07
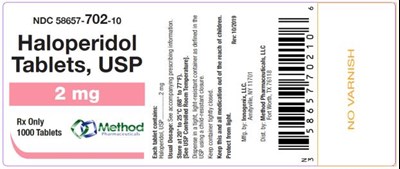
This is a description of a medication called Haloperidol in tablet form. The NDC number for the medication is 58657-702-10 and it is only available with a prescription. The text also includes the quantity of the tablets, which is 1000. Overall, this appears to be a labeling or packaging detail for the medication.*
PRINCIPAL DISPLAY PANELNDC 58657-703-01Haloperidol Tablets, USP5 mgRx Only100 Tablets - haloperidol tablets usp 08

This is a medication description for Haloperidol Tablets, USP. It includes the NDC code, 58657-703-01, the drug name, Haloperidol and the form, tablets. The quantity is also mentioned with 100 tablets. No further information is provided.*
PRINCIPAL DISPLAY PANELNDC 58657-703-10Haloperidol Tablets, USP5 mgRx Only1000 Tablets - haloperidol tablets usp 09
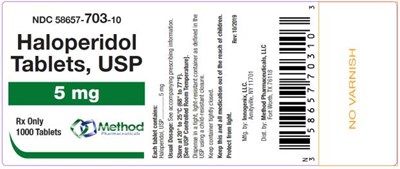
This is a medication with the NDC code 58657-703-10 that contains Haloperidol in tablet form.*
PRINCIPAL DISPLAY PANELNDC 58657-704-01Haloperidol Tablets, USP10 mgRx Only100 Tablets - haloperidol tablets usp 0a
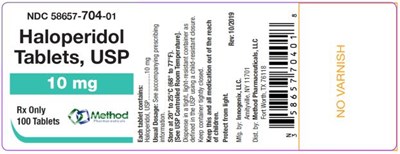
Haloperidol Tablets, USP are prescription medicine indicated for the treatment of schizophrenia. The NDC (National Drug Code) for this medication is 58657-704-01. The remaining text appears to be incomplete or unclear.*
PRINCIPAL DISPLAY PANELNDC 58657-704-10Haloperidol Tablets, USP10 mgRx Only1000 Tablets - haloperidol tablets usp 0b

This is a medication label for Haloperidol Tablets. The National Drug Code is 58657-704-10 and the medication is available in a container of 1000 tablets. The tablets contain Methy and are recognized by the USP (United States Pharmacopeia). It is marked as a prescription-only medication.*
PRINCIPAL DISPLAY PANELNDC 58657-705-01Haloperidol Tablets, USP20 mgRx Only100 Tablets - haloperidol tablets usp 0c

The text describes a medication with NDC code 58657-705-01 called Haloperidol in tablet form. The medication is only available on prescription and comes in a container of 100 tablets. Additionally, there is a revision number of 102119 mentioned, and the tablets are to be stored in a protective manner.*
PRINCIPAL DISPLAY PANELNDC 58657-705-10Haloperidol Tablets, USP20 mgRx Only1000 Tablets - haloperidol tablets usp 0d

This is a medication called Haloperidol, which comes in the form of tablets. The NDC number for this medication is 58657-705-10. There is some additional text that is blurry and difficult to read, but it appears to reference the name "Reonly" and possibly the initials GLWMethodt.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
