Product Images Diclofenac Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Diclofenac Sodium NDC 67296-1480 by Redpharm Drug, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
label 50 count 100 - 50mg label

This is a label for Diclofenac Sodium tablets, which come in a delayed-release formulation. The NDC number is 68001-280-00 and the label includes instructions for pharmacists to provide a medication guide to each patient with their prescription. There is also a section on the label designed for batch coding and a 2D barcode. The dimensions of the label are 45mm x 145mm.*
label 75 count 100 - 75mg label
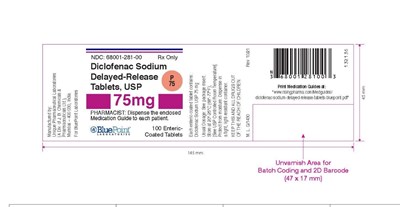
This is a description of Diclofenac Sodium Delayed-Release Tablets by BluePoint Laboratories. The NDC code is 68001-281-00, and it is recommended for prescription use only. The medication guide should be dispensed to each patient. The usual dosage should be seen in the package insert, and the tablets should be stored between 20-25°C. The batch coding and 2D barcode area is approximately 47 x 17 mm.*
label 25 count 100 - image 01
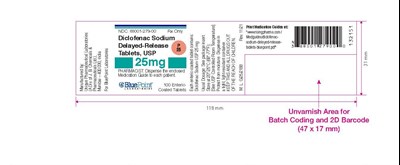
The text seems to be a medication guide for Diclofenac Sodium Delayed-Release Tablets. It contains information on the dosage, NDC number, and instructions for pharmacists to dispense the medication with an enclosed medication guide to patients. The text also mentions a website to print additional medication guides. Additionally, it includes information on a batch code and 2D barcode.*
label 50 count 100 - image 02

This is the label of a medicine container with the name Diclofenac Sodium delayed-release tablets. It contains 100 enteric-coated blue tablets. The size of each tablet is 45mm. The label also includes a batch coding and 2D barcode area. The medication guide is included in the package and should be given to each patient by the pharmacist.*
label 25 count 100 - image 03

This is a medication package of Diclofenac Sodium Delayed-Release Tablets, USP. It contains a medication guide that is to be dispensed to each patient. The package has a barcode area for batch coding. NDC number for this medication is 68001-281-00.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

