Product Images Spironolactone
View Photos of Packaging, Labels & Appearance
- EPM 30 COUNT - 67296 1692 3
- Image - b9a028ee 5720 4b9e a625 f7130aafd084 01
- Image - b9a028ee 5720 4b9e a625 f7130aafd084 02
- Image - b9a028ee 5720 4b9e a625 f7130aafd084 03
- image4 - b9a028ee 5720 4b9e a625 f7130aafd084 04
- image5 - b9a028ee 5720 4b9e a625 f7130aafd084 05
- image6 - b9a028ee 5720 4b9e a625 f7130aafd084 06
Product Label Images
The following 7 images provide visual information about the product associated with Spironolactone NDC 67296-1692 by Redpharm Drug, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
EPM 30 COUNT - 67296 1692 3

This is a description of a medication: Spironolactone 25mg tablets manufactured by Cadila Healthcare LTD in Baddi, India. The NDC number is 67296-1692-3 and the product contains 30 tablets, with an expiration date of 11/20 and lot number 8D4647X1. The drug is distributed by Redpharm Drug, located in Eden Prairie, MN. The recommended storage temperature is 2025 C (68-77 F). Adult dosage information is available in the package insert.*
Image - b9a028ee 5720 4b9e a625 f7130aafd084 02
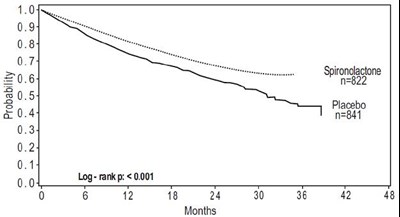
The text is presenting results of a study or trial involving the use of Spironolactone and Placebo in two different groups represented by n=822 and n=841. The study involved monitoring the groups for up to 42 months as represented on the timeline. The results of the study were significant with a log-rank p<0.001. No further information is available from the given text.*
Image - b9a028ee 5720 4b9e a625 f7130aafd084 03
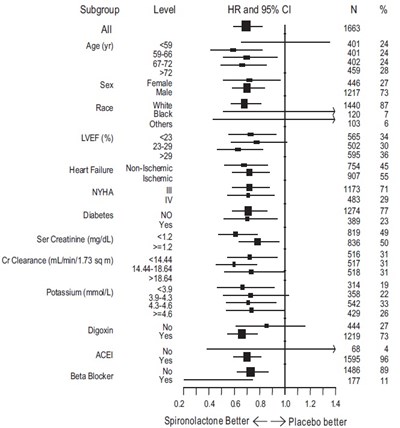
This is a table showing the various subgroups of a study related to heart failure. The subgroups are based on age, sex, race, LVEF (%), NYHA, Diabetes, Serum creatinine (mg/dL), CrClearance (mLimin/1.73 sqm), Potassium (mmoilL), Digoxin, ACE, and Beta Blocker. The table also includes the number of participants in each subgroup along with their HR (Heart Rate) and 95% CI (Confidence Interval) N (Sample Size) values. Finally, there is a comparison of the effectiveness of Spironolactone and Placebo.*
image4 - b9a028ee 5720 4b9e a625 f7130aafd084 04

NDC number 68382-660-01 belongs to a Spironolactone drug in tablet form. Each tablet is film-coated and contains 25mg of Spironolactone, a medication used for treating various heart, liver, and kidney-related health conditions. The ideal dosage is mentioned in the package insert. It should be stored at room temperature and protected from light. This drug should be kept out of children's reach. It is manufactured in Baddi, India, by the Cadia Healthcare Ltd., and distributed by Zydus Pharmaceuticals (USA) Inc. located in Pennington, NJ.*
image5 - b9a028ee 5720 4b9e a625 f7130aafd084 05

This is a description of a medication called Spironolactone which comes in the form of tablets. Each film coated tablet contains 50 mg of Spironolactone, USP for prescribed use. The packaging and usage instructions are provided in the package insert. The medication should be stored at room temperature and protected from light. It should be dispensed in a tight, light-resistant container to ensure quality. This drug should be kept out of reach of children. The drug is manufactured by Cadila Healthcare Ltd. in Badd, India, and distributed by Zydus Pharmaceuticals (USA) Inc. in Pennington, NJ.*
image6 - b9a028ee 5720 4b9e a625 f7130aafd084 06

NDC 68382-662-01 is a prescription drug called Spironolactone that comes in the form of tablets. Each film-coated tablet contains 100 mg of spironolactone, USP. The package insert should be consulted for complete prescribing information, including usual dosage. It should be stored in a light-resistant container within the USP controlled room temperature of 20° to 25°C (68° to 77°F). Manufactured by Cadia Healthcare Ltd. in Baddi, India, and distributed by Zydus Pharmaceuticals (USA) Inc. in Pennington, NJ 08534. It is advised to keep this and all drugs out of the reach of children.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
