Product Images Carvedilol
View Photos of Packaging, Labels & Appearance
- Carvedilol Tablets USP, 12.5 mg - NDC 72888-036-00 - 1000 Tablets Container Label - carvedilol tab usp 12pt5mg 1000s label
- Carvedilol Tablets USP, 12.5 mg - NDC 72888-036-01 - 100 Tablets Container Label - carvedilol tab usp 12pt5mg 100s label
- Carvedilol Tablets USP, 12.5 mg - NDC 72888-036-30 - 30 Tablets Container Label - carvedilol tab usp 12pt5mg 30s label
- Carvedilol Tablets USP, 12.5 mg - NDC 72888-036-05 - 500 Tablets Container Label - carvedilol tab usp 12pt5mg 500s label
- Carvedilol Tablets USP, 25 mg - NDC 72888-037-00 - 1000 Tablets Container Label - carvedilol tab usp 25mg 1000s label
- Carvedilol Tablets USP, 25 mg - NDC 72888-037-01 - 100 Tablets Container Label - carvedilol tab usp 25mg 100s label
- Carvedilol Tablets USP, 25 mg - NDC 72888-037-30 - 30 Tablets Container Label - carvedilol tab usp 25mg 30s label
- Carvedilol Tablets USP, 25 mg - NDC 72888-037-05 - 500 Tablets Container Label - carvedilol tab usp 25mg 500s label
- Carvedilol Tablets USP, 3.125 mg - NDC 72888-034-00 - 1000 Tablets Container Label - carvedilol tab usp 3pt125mg 1000s label
- Carvedilol Tablets USP, 3.125 mg - NDC 72888-034-01 - 100 Tablets Container Label - carvedilol tab usp 3pt125mg 100s label
- Carvedilol Tablets USP, 3.125 mg - NDC 72888-034-30 - 30 Tablets Container Label - carvedilol tab usp 3pt125mg 30s label
- Carvedilol Tablets USP, 3.125 mg - NDC 72888-034-05 - 500 Tablets Container Label - carvedilol tab usp 3pt125mg 500s label
- Carvedilol Tablets USP, 6.25 mg - NDC 72888-035-00 - 1000 Tablets Container Label - carvedilol tab usp 6pt25mg 1000s label
- Carvedilol Tablets USP, 6.25 mg - NDC 72888-035-01 - 100 Tablets Container Label - carvedilol tab usp 6pt25mg 100s label
- Carvedilol Tablets USP, 6.25 mg - NDC 72888-035-30 - 30 Tablets Container Label - carvedilol tab usp 6pt25mg 30s label
- Carvedilol Tablets USP, 6.25 mg - NDC 72888-035-05 - 500 Tablets Container Label - carvedilol tab usp 6pt25mg 500s label
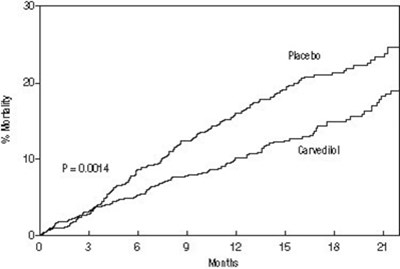
- Figure 1. Survival Analysis for COPERNICUS (intent-to-treat) - carvedilol tab usp figure1
- Figure 2. Effects on Mortality for Subgroups in COPERNICUS - carvedilol tab usp figure2
- Figure 3. Survival Analysis for CAPRICORN (intent-to-treat) - carvedilol tab usp figure3
- Figure 4. Effects on Mortality for Subgroups in CAPRICORN - carvedilol tab usp figure4
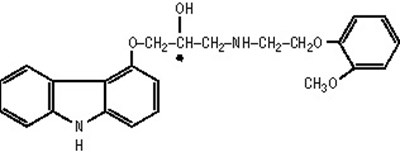
- carvedilol chemical structure - carvedilol tab usp structure
Product Label Images
The following 21 images provide visual information about the product associated with Carvedilol NDC 67296-1900 by Redpharm Drug, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Carvedilol Tablets USP, 12.5 mg - NDC 72888-036-00 - 1000 Tablets Container Label - carvedilol tab usp 12pt5mg 1000s label

Carvedilol Tablets USP, 12.5 mg - NDC 72888-036-01 - 100 Tablets Container Label - carvedilol tab usp 12pt5mg 100s label

Carvedilol Tablets USP, 12.5 mg - NDC 72888-036-30 - 30 Tablets Container Label - carvedilol tab usp 12pt5mg 30s label

Carvedilol Tablets USP, 12.5 mg - NDC 72888-036-05 - 500 Tablets Container Label - carvedilol tab usp 12pt5mg 500s label

Each tablet contains carvedilol, a medication used for treating high blood pressure and heart failure. The tablets are available in a strength of 12.5mg. It is important to store the medication below 30°C (86°F) and protect it from moisture. The tablets should not be accepted if the seal under the cap is missing or broken. It should be dispensed in a tight, light-resistant container. The package contains 500 tablets and is for prescription use only. The dosage instructions can be found in the accompanying prescribing information. The medication is distributed by Advagen Pharma Limited, located at 666 Plainsboro Road Suite 605, Plainsboro, NJ, 08536, USA. It is manufactured by Rubicon Research Private Limited in Ambernath, Dist. Thane, 421506, India. The manufacturing license number is KD-682 21144.*
Carvedilol Tablets USP, 25 mg - NDC 72888-037-00 - 1000 Tablets Container Label - carvedilol tab usp 25mg 1000s label

This text appears to be the label information of a medication called carvedilol. Each tablet contains 25 mg of carvedilol and is identified by NDC code 72888-037-00. The tablets should be stored below 30°C (86°F) and protected from moisture. It is advised not to accept the medication if the seal under the cap is missing or broken. The tablets should be dispensed in a tight, light-resistant container. For dosage instructions, refer to the accompanying prescribing information. The product is distributed by Advagen Pharma Limited, located at 666 Plainsboro Road Suite 605, Plainsboro, NJ, 08536, USA. It is manufactured by Rubicon Research Private Limited in Ambernath, Dist. Thane, 421506, India. The manufacturing license number is KD-68221149. The indicated area for overprinting is 60 mm x 25 mm.*
Carvedilol Tablets USP, 25 mg - NDC 72888-037-01 - 100 Tablets Container Label - carvedilol tab usp 25mg 100s label

Each tablet contains carvedilol, 25 mg. Store below 30°C (86°F). Protect from moisture. Do not accept if seal under cap is missing or broken. Dispense in a tight, light-resistant container. 100 Tablets. Rx Only. Dosage: See accompanying prescribing information. Distributed by Advagen Pharma Limited, Plainsboro, NJ, 08536, USA. Manufactured by Rubicon Research Private Limited, Ambernath, Dist. Thane, 421506, India. Mfg Lic No.: KD-682 21147. Overprinting Area: 45 mm X 20 mm.*
Carvedilol Tablets USP, 25 mg - NDC 72888-037-30 - 30 Tablets Container Label - carvedilol tab usp 25mg 30s label

The description is not available.*
Carvedilol Tablets USP, 25 mg - NDC 72888-037-05 - 500 Tablets Container Label - carvedilol tab usp 25mg 500s label

Each tablet contains 25mg of carvedilol. The tablets should be stored below 30°C (86°F) and protected from moisture. The product should not be accepted if the seal under the cap is missing or broken. It should be dispensed in a tight, light-resistant container. The package contains 500 tablets and is for prescription use only. Dosage information can be found in the accompanying prescribing information. The product is distributed by Advagen Pharma Limited, located at 666 Plainsboro Road Suite 605, Plainsboro, NJ, 08536, USA. It is manufactured by Rubicon Research Private Limited in Ambernath, Dist. Thane, 421506, India.*
Carvedilol Tablets USP, 3.125 mg - NDC 72888-034-00 - 1000 Tablets Container Label - carvedilol tab usp 3pt125mg 1000s label

This text appears to be the label or packaging information for a medication called Carvedilol. Each tablet contains 3.125 mg of the active ingredient. It should be stored below 30°C (86°F) and protected from moisture. The tablets should not be accepted if the seal under the cap is missing or broken. The dosage instructions can be found in the accompanying prescribing information. The text also includes information about the distributor, Advagen Pharma Limited, located in Plainsboro, NJ, USA. The manufacturer is Rubicon Research Private Limited, located in India. Unfortunately, the remaining part of the text is not readable.*
Carvedilol Tablets USP, 3.125 mg - NDC 72888-034-01 - 100 Tablets Container Label - carvedilol tab usp 3pt125mg 100s label

Carvedilol Tablets USP, 3.125 mg - NDC 72888-034-30 - 30 Tablets Container Label - carvedilol tab usp 3pt125mg 30s label

Each tablet contains carvedilol, a medication used to treat heart failure and high blood pressure. The tablets come in a pack of 30 and should be stored in a tight and light-resistant container below 30°C (86°F) to protect them from moisture. The dosage should be determined based on the accompanying prescribing information. The tablets are distributed by Advagen Pharma Limited in Plainsboro, NJ, USA. They are manufactured by Rubicon Research Private Limited in Ambernath, India. It is important to check the seal under the cap before accepting the medication, as it should not be taken if the seal is missing or broken.*
Carvedilol Tablets USP, 3.125 mg - NDC 72888-034-05 - 500 Tablets Container Label - carvedilol tab usp 3pt125mg 500s label

Carvedilol Tablets USP, 6.25 mg - NDC 72888-035-00 - 1000 Tablets Container Label - carvedilol tab usp 6pt25mg 1000s label

Each tablet of Carvedilol contains 6.25 mg of the active ingredient. These tablets should be stored below 30°C (86°F) and protected from moisture. It is important not to use the tablets if the seal under the cap is missing or broken. The tablets should be dispensed in a tightly sealed, light-resistant container. The dosage instructions should be found in the accompanying prescribing information. These Carvedilol tablets are distributed by Advagen Pharma Limited and come in a pack of 1000 tablets. This medication is only available with a prescription. The manufacturing of these tablets is done by Rubicon Research Private Limited in India. The manufacturing license number is KD-682.*
Carvedilol Tablets USP, 6.25 mg - NDC 72888-035-01 - 100 Tablets Container Label - carvedilol tab usp 6pt25mg 100s label

Carvedilol Tablets USP, 6.25 mg - NDC 72888-035-30 - 30 Tablets Container Label - carvedilol tab usp 6pt25mg 30s label

Carvedilol Tablets USP, 6.25 mg - NDC 72888-035-05 - 500 Tablets Container Label - carvedilol tab usp 6pt25mg 500s label

Each tablet contains carvedilol 6.25 mg. This medication should be stored below 30°C (86°F) and protected from moisture. It is important not to accept the medication if the seal under the cap is missing or broken. The tablets should be dispensed in a tight, light-resistant container. The dosage information can be found in the accompanying prescribing information. The product is distributed by Advagen Pharma Limited and manufactured by Rubicon Research Private Limited in India. The package contains 500 tablets.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.