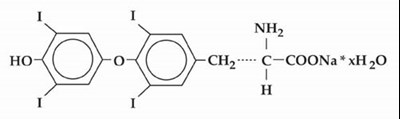
Product Images Levothyroxine Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Levothyroxine Sodium NDC 67296-1908 by Redpharm Drug, Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
label-112-mcg-90-tab - levothyroxine 112 mcg 90 tab

Each tablet contains 112 micrograms of levothyroxine sodium. Store the tablets between 20°C to 25°C (68°F to 77°F), with permitted excursions between 15°C to 30°C (59°F to 86°F). Protect the tablets from light and moisture. Dispense in a tight, light-resistant container. Do not use if the seal over the bottle opening is broken or missing. The tablets are manufactured by Intas Pharmaceuticals Limited in Dehradun, India, for Accord Healthcare, Inc. Each bottle contains 90 tablets. The product is labeled as Levothyroxine Sodium Tablets, USP. The National Drug Code (NDC) for the product is 16729-452-15. The lot number and expiry date (EXP) can be found on the blank and varnish-free area of the packaging.*
label-125-mcg-90-tab - levothyroxine 125 mcg 90 tab

Each tablet contains 125 meg (0.125 mg) of levothyroxine sodium USP. This medication is used to treat an underactive thyroid (hypothyroidism). It is important to store these tablets at a temperature between 20°C to 25°C (68°F to 77°F) with permissible excursions to 15°C to 30°C (59°F to 86°F). The tablets should be protected from light and moisture. The manufacturing company is Intas Pharmaceuticals Limited located in Dehradun, India. This medication is available by prescription only and is manufactured for Accord Healthcare, Inc. Please refer to the package insert for full prescribing information.*
label-200-mcg-90-tab - levothyroxine 200 mcg 90 tab

Each tablet contains 200 meg (0.2 mg) of levothyroxine sodium USP. The tablets should be stored at a temperature between 20°C to 25°C (68°F to 77°F), with excursions permitted to 15°C to 30°C (59°F to 86°F). It is important to protect the tablets from light and moisture. The tablets should be dispensed in a tight, light-resistant container as described in the USP. Do not accept the tablets if the seal over the bottle opening is broken or missing. The tablets are manufactured by Accord Healthcare, Inc. and Intas Pharmaceuticals Limited.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.