Product Images Tosymra
View Photos of Packaging, Labels & Appearance
- Blister Lidding Artwork - Blister Lidding Artwork
- Carton Artwork - Carton Artwork
- Fig-A - FigureA
- Fig-B - FigureB
- Fig-C - FigureC
- Fig-D - FigureD
- Fig-E - FigureE
- Fig-F - FigureF
- Fig-G - FigureG
- Fig-H-I - FigureHI
- Fig-J - FigureJ
- Fig-K - FigureK
- Fig-L - FigureL
- Fig-M - FigureM
- Table 1 - Table 1
- Table 2 - Table 2
- Table 3 - Table 3
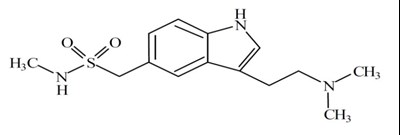
- Structure - structure
Product Label Images
The following 18 images provide visual information about the product associated with Tosymra NDC 67857-812 by Promius Pharma, Llc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Blister Lidding Artwork - Blister Lidding Artwork

TOSYMRA is a prescription medication containing 10mg of sumatriptan. It is intended for intranasal use only and has a warning to keep away from children. The medication should not be stored in a refrigerator or freezer and should be kept between 20°C to 25°C.*
Fig-A - FigureA
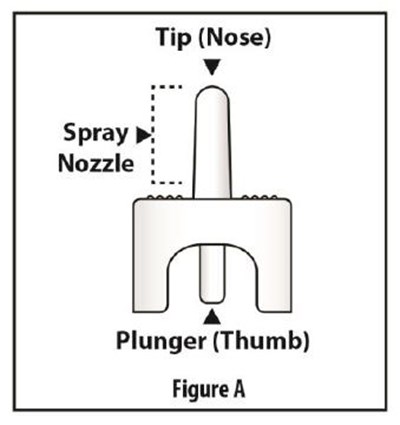
The text describes a figure labeled as "Figure A" consisting of a tip or nose and a plunger or thumb. This may be part of an instructional manual or diagram for using a tool or medical device.*
Fig-K - FigureK

This text may describe a set of instructions for using a medical device or tool. The instructions involve breathing in and pressing a plunger up, as shown in Figure K.*
Fig-L - FigureL
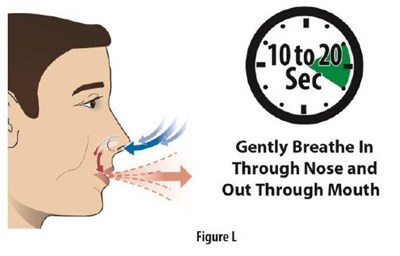
The text appears to be a set of instructions for breathing exercise, asking the reader to breathe in gently through their nose and exhale slowly through their mouth. Figure L is also mentioned, but without context it is unclear what it represents.*
Table 1 - Table 1
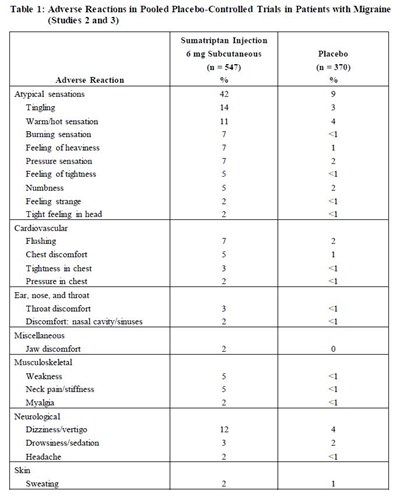
This is a table showing adverse reactions in patients with migraines who received Sumatriptan Injection and Placebo in two studies. The table presents the percentage of patients who experienced different adverse reactions. The adverse reactions are categorized into Atypical sensations, Cardiovascular, Ear, nose, and throat, Miscellaneous, Musculoskeletal, Neurological, and Skin. The adverse reactions reported in the Sumatriptan Injection group include tingling, warm/hot sensation, burning sensation, and dizziness/vertigo, among others. The placebo group reported fewer adverse reactions, primarily atypical sensations.*
Table 2 - Table 2

This is a table showing the proportion of patients with relief from migraines and the incidence of adverse reactions based on the time and dosage of sumatriptan used in Study 1. The table includes data for different doses of sumatriptan ranging from 1mg to 6mg and indicates the percentage of patients with relief at different time intervals, as well as the incidence of adverse reactions. Relief is defined as a reduction of moderate or severe pain to no or mild pain after dosing without the use of rescue medication. The efficacy of Tosymra nasal spray was based on bioavailability to 4 mg sumatriptan SC injection.*
Table 3 - Table 3
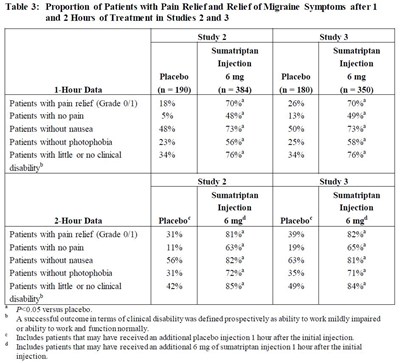
Table 3 summarizes the results of two studies evaluating the proportion of patients experiencing pain relief and relief of migraine symptoms after 1 and 2 hours of treatment with sumatriptan injection and placebo. The data includes the percentage of patients with pain relief, no pain, no nausea, and no photophobia, as well as the percentage of patients with little or no clinical disability. Overall, patients treated with sumatriptan experienced greater pain relief and symptom relief compared to those who received placebo. These results suggest that sumatriptan injection may be an effective treatment option for migraine.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.