Product Images Zaleplon
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Zaleplon NDC 67877-210 by Ascend Laboratories, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Zaleplon Capsule 10 mg - Container Label - zal 10mg cap cnt lbl

Each capsule of the medicine contains 10 mg Zaleplon. The medicine is marketed by ATEPLONG located in Montvale, NJ and is stored at a controlled room temperature of 20° to 25°C (68°to 77°F). The pharmacist must dispense the medicine in a light-resistant container as defined in the USP. The medicine is available in a pack of 100 capsules and must be dispensed with the medication guide.*
Zaleplon Capsules 5 mg - Container Label - zal 5mg cap cont lbl

Each capsule of Zaleplon contains 5 mg of the medication. The usual dosage should be checked from the accompanying information. The capsule has FD&C Yellow No.5 (tartazine) which is a color additive. It must be stored in a room with controlled temperature, ideally between 20° to 25°C (68 to 77°F) [See USP], and must be dispensed in a light-resistant container as defined in the USP. It is manufactured by Unichem Laboratories Ltd in India and marketed by Ascond Laboratories in New Jersey. The NDC code for the medication is 67877-210-30, and it comes in a bottle of 30 capsules. Pharmacists should dispense the medicine with the medication guide, and it is only available through prescription (Rx only).*
Zaleplon Strcture - zal struct
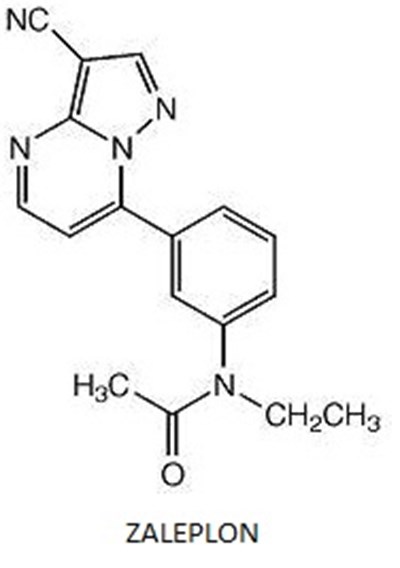
The text contains chemical formulas and a drug name. It seems to be related to chemistry and pharmacology. The drug name is "Zaleplon".*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.