Product Images Telmisartan
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Telmisartan NDC 67877-482 by Ascend Laboratories, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
a70f59be 00af 4225 8711 3c7fb54c2430 04

ASCEND is a medication containing 20mg of telmisartan USP per tablet. These moisture-sensitive tablets are distributed by Ascend Laboratories and manufactured by P Inventia Healthcare Limited in India. Each bottle contains 30 tablets to be stored at 20°C - 25°C. Patients should not remove tablets from the bottle until immediately before administering. A patient information leaflet is enclosed, and prescription is required.*
a70f59be 00af 4225 8711 3c7fb54c2430 06
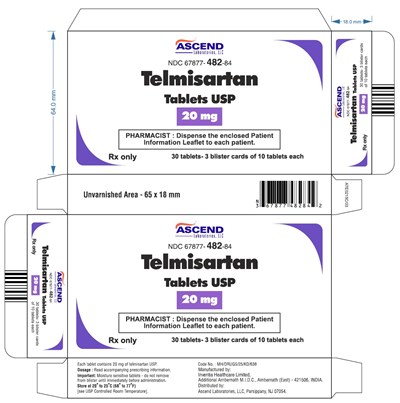
This is a text related to medication. The name of the medication is "Telmisartan Tablets USP", which comes in a bottle containing 30 tablets. The tablets are moisture-sensitive and should not be removed from the blister until immediately before administration. The pharmacist is recommended to dispense Patient Information Leaflet to each patient. The manufacturer is Inventia Healthcare Limited, located in Ambernath, India. The medication is distributed in the United States by Ascend Laboratories, LLC, located in Parsippany, NJ. The text also includes a warning about the sensitivity of the tablets to moisture and the storage instructions for the medication. There is no useful information about the measurements mentioned in the first lines.*
a70f59be 00af 4225 8711 3c7fb54c2430 07

This is a drug label for Telmisartan Tablets USP. The tablets are moisture sensitive and each tablet contains 40mg of telmisartan USP. The drug should not be removed from the bottle until immediately before administration. Patients should read the enclosed patient information leaflet. The tablets should be stored at 20° to 25°C (68° to 77°F) and the prescribing information should be followed carefully. The drug is manufactured by Inventia Healthcare Limited, in India, and distributed by Ascend Laboratories, LLC, in Parsippany, NJ. There is an unvarnished area on the label that measures 45 x 15 mm.*
a70f59be 00af 4225 8711 3c7fb54c2430 09
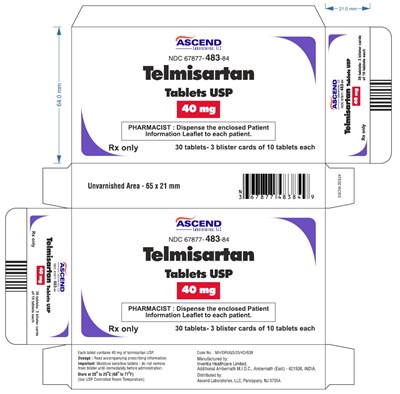
This is a description of a medication called Telmisartan Tablets USP produced by Ascend Laboratories. It is available in 30 tablets, divided into three blister cards, and is recommended only with a prescription. The pharmacists are instructed to provide the patient with the enclosed patient information leaflet. The dosage instructions are mentioned as a separate document. The medication should be stored between 20 to 25°C (68 to 77°F).*
a70f59be 00af 4225 8711 3c7fb54c2430 10

ASCEND is a medication used to treat hypertension containing 30 tablets with each tablet containing 80 mg of Telmisartan USP. It is manufactured by Inventia Healthcare Limited and it comes with packaging information for pharmacist and patients to follow. It should be stored between 20-25°C and is distributed by Ascend Laboratories, LLC.*
a70f59be 00af 4225 8711 3c7fb54c2430 12
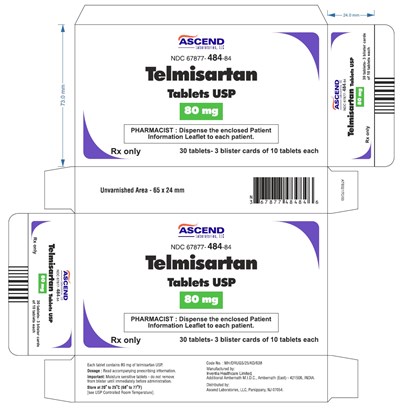
This is a medication information leaflet for Telmisartan Tablets USP from Ascend Laboratories. Each table contains 80 mg of Telmisartan USP. The tablets are moisture sensitive, so they should not be removed from the blister until immediately before administration. They should be stored at 20-25°C (68-77°F). The lot number of the medication is MH/DRUGS/25/KD/638. The barcode number is NDC 67877-484-84. The size of the unvarnished area is 65 x 24 mm. There are 30 tablets in total, grouped into 3 blister cards of 10 tablets each. The text contains several gibberish strings and letters produced by .*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.






