Product Images Sildenafil Citrate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Sildenafil Citrate NDC 67877-648 by Ascend Laboratories, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
sildenafil cart 2

Sildenafil for Oral Suspension is a grape-flavored medicine that should be shaken well before use. The bottle contains 112 ml following reconstitution. Each milliliter of suspension contains 10 mg of sildenafil, to be reconstituted with 23 ml of water. The medicine should be stored at a temperature below 30°C. If refrigerated, keep it between 2°C-8°C. This medicine should not be frozen and should be discarded within 60 days after reconstitution. The text also includes information about the manufacturer and distributor, as well as instructions for reconstitution.*
sildenafil cont 2

This is a medication that needs to be stored below 30°C (86°F) and protected from moisture. After reconstitution, it can be stored in the refrigerator at 2-8°C (36° - 46°F). Each bottle contains 1.57 g of sildenafi citrate which contains 10 mg of sidenaf (as citrate) when reconstituted with 93 mL water. The recommended dosage is not available, hence the user should see prescribing information. The medication is grape-flavored and needs to be shaken well before each use. It is manufactured in Mumbai, India by Alkem Laboratories Ltd. and distributed by Ascend Laboratories, LLC in Parsippany, NJ. The medication comes in 112 mL following reconstitution. The label has a non-coated area of 45 x 30 mm (LXH), and the rest of the label has UV Varnish.*
sildenafil-fig7 - sildenafil fig7

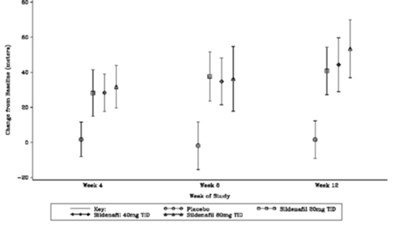
This text appears to be a mix of medicine names and dosages, therefore likely presenting potential drug interactions and their effects. It also includes a chart that shows the change relative to the use of Sildenafil alone and mentions that no exercise capacity benefit is expected when Sildenafil is added to Bosentan therapy based on clinical studies.*
sildenafil-fig8 - sildenafil fig8

This is a table with information on the interaction of various drugs with other interacting drugs. The table includes the drug name and various measurements such as INR, AUC, and Cmax. The data appears to be related to changes in drug effectiveness when combined with other drugs. There is a note at the end of the table regarding the lack of benefit of adding Sidenafi to Bosentan therapy.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.