Product Images Sacubitril And Valsartan
View Photos of Packaging, Labels & Appearance
- sacu-fig-a-1 - sacu fig a 1
- sacu-fig-b-1 - sacu fig b 1
- sacu-fig-c-2 - sacu fig c 2
- sacu-figure-4-c - sacu figure 4 c
- sacu-figure1 - sacu figure1
- sacu-figure2 - sacu figure2
- sacu-figure5-a - sacu figure5 a
- sacu-figure6-a - sacu figure6 a
- sacu-figure7 - sacu figure7
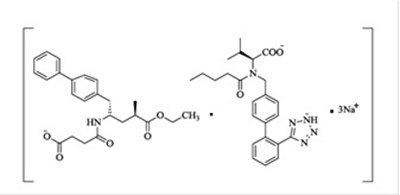
- sacu-structure - sacu structure
- sacu-val-24-26-180tab-a - sacu val 24 26 180tab a
- sacu-val-24-26-60tab-a - sacu val 24 26 60tab a
- sacu-val-49-51-180tab-a - sacu val 49 51 180tab a
- sacu-val-49-51-60tab-a - sacu val 49 51 60tab a
- sacu-val-97-103-180tab-a - sacu val 97 103 180tab a
- sacu-val-97-103-60tab-a - sacu val 97 103 60tab a
Product Label Images
The following 16 images provide visual information about the product associated with Sacubitril And Valsartan NDC 67877-709 by Ascend Laboratories, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
sacu-fig-a-1 - sacu fig a 1
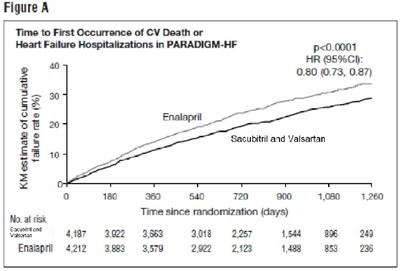
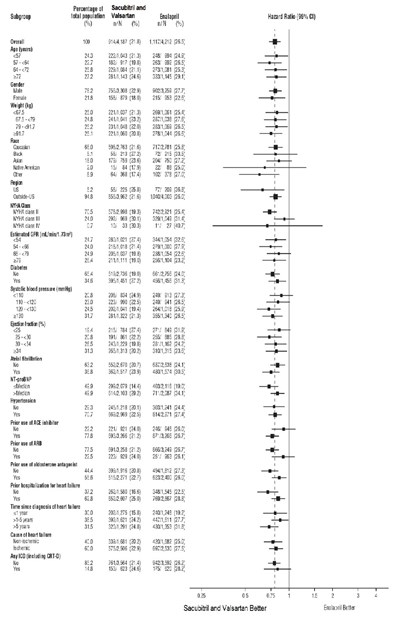
The text provides data related to the Time to First Occurrence of CV Death or Heart Failure Hospitalizations in a study called PARADIGM-HF. The results show a statistically significant difference (p<0.0001) between the group treated with Sacubitril and Valsartan compared to the group treated with Enalapril. The Hazard Ratio (HR) with 95% confidence interval was 0.80 (0.73, 0.87), indicating a lower risk in the Sacubitril and Valsartan group. The data is presented over various time intervals since randomization (in days) for both treatment groups. This information can be helpful for evaluating the effectiveness of these treatments in reducing cardiovascular events and heart failure hospitalizations.*
sacu-fig-b-1 - sacu fig b 1

This text provides information about the time to occurrence of cardiovascular death in the PARADIGM-HF study. It displays a graph showing the hazard ratio (HR) of 0.80 with a 95% confidence interval of 0.71 to 0.89. The graph also includes the percentage of patients on Sacubitril and Valsartan, with time intervals labeled in days. The study involves two groups: one receiving Sacubitril and Valsartan, and the other receiving Enalapril. The number of patients at risk and the time since randomization in days are also shown in the data.*
sacu-fig-c-2 - sacu fig c 2
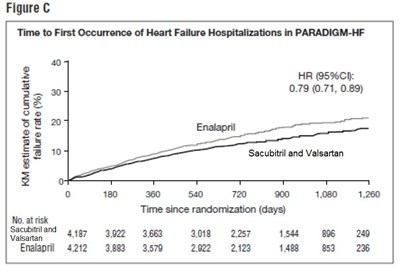
The provided text seems to be a figure related to the "Time to First Occurrence of Heart Failure Hospitalizations in PARADIGM-HF" study. It shows the Hazard Ratio (HR) with 95% Confidence Interval (CI) for the comparison between Sacubitril and Valsartan versus Enalapril. The time since randomization is depicted in days along with the number of patients at risk at different timepoints. This figure provides insight into the time taken for the first occurrence of heart failure hospitalizations in the PARADIGM-HF study for the two treatments being compared.*
sacu-figure1 - sacu figure1
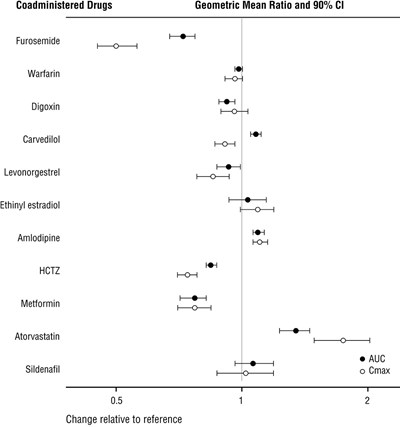
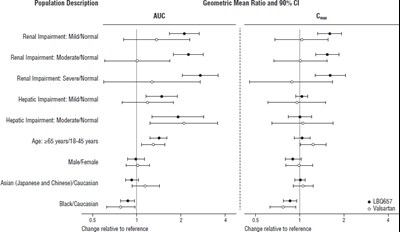
This text seems to list a variety of drugs that could potentially be coadministered. The list includes drugs such as Furosemide, Warfarin, Digoxin, Carvedilol, Levonorgestrel, Ethinyl estradiol, Amlodipine, HCTZ, Metformin, Atorvastatin, and Sildenafil. The text also mentions Geometric Mean Ratio and 90% Confidence Interval. These drugs may have interactions or effects when taken together, so it is essential to consider these factors when prescribing medication to patients.*
sacu-val-24-26-180tab-a - sacu val 24 26 180tab a

This text provides information about Sacubitril and Valsartan Tablets with a dosage of 24 mg/26 mg. It includes instructions on storage conditions, dosage details, and a caution to keep the tablets out of the reach of children. The tablets are manufactured by Akem Laboratories Ltd in India and distributed by A Laboratories. The provided NDC number is 67877-707-51.*
sacu-val-49-51-180tab-a - sacu val 49 51 180tab a

Each film coated tablet contains 49 mg of sacubitril equivalent to 51.2 mg of sacubitril, monosodium, and 51 mg of valsartan, equivalent to 56 mg of valsartan disodium. The recommended dosage should be in the range of 20°C to 25°C (68°F to 77°F) for Valsartan Tablets. It is important to control the temperature within 15°C to 30°C (59°F to 86°F) and protect the tablets from moisture. The tablets should be kept in a dry place and away from the reach of children. A label with UV Varnish is advisable. These tablets are manufactured by Alkem Laboratories Ltd. in Mumbai, India, and distributed by Ascend Laboratories, LLC in Parsippany, NJ.*
sacu-val-49-51-60tab-a - sacu val 49 51 60tab a

This text provides information about Sacubitril and Valsartan tablets, including their composition, dosage instructions, storage recommendations, and manufacturer details. The tablets contain 49 mg of Sacubitril and 51 mg of Valsartan. The typical dosage should be determined by a healthcare provider. The storage conditions suggest keeping the tablets between 20°C to 25°C (65°F to 77°F) with excursions permitted between 15°C to 30°C (59°F to 86°F). The manufacturer is listed as Akem Laboratories, based in Mumbai, India. The tablets are distributed by A Liboaioes, LG headquartered in Parsippany, NJ. Each box contains 60 tablets.*
sacu-val-97-103-180tab-a - sacu val 97 103 180tab a

Each film-coated tablet contains 97 mg of sacubtr equivalent to 102.4 mg of sacubltrilmonosodium and 103 mg of valsartan equivalent to 1132 mg of valsartan disodium. The usual dosage is recommended in the prescribing information. It is advised to store the tablets at 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C to 30°C (59°F to 86°F) to protect from moisture. Always keep this medication and all drugs out of the reach of children. This product is manufactured by Alkem Laboratories in Mumbai, India, and distributed by Ascend Laboratories, LLC in Parsippany, NJ. Each pack of the tablets contains 180 tablets.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.