Product Images Everolimus
View Photos of Packaging, Labels & Appearance
Product Label Images
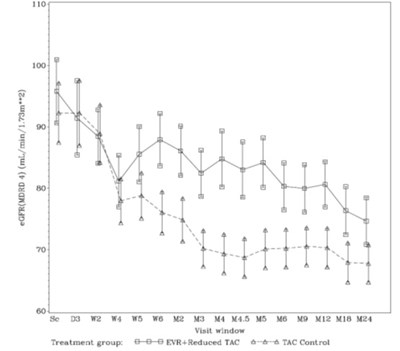
The following 7 images provide visual information about the product associated with Everolimus NDC 67877-720 by Ascend Laboratories, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
evero-bottle-1mg-60s - evero bottle 1mg 60s

Each tablet of Everolimus contains an unspecified amount of the drug. It is recommended to refer to the package insert for the proper dosage. The tablets should be stored at a temperature not exceeding 25°C (77°F) but may be kept between 15°C to 30°C (59°F to 86°F). The medication should be protected from light and moisture. In case of side effects, call a doctor or report to the FDA. Everolimus Tablets are manufactured by Alkem Laboratories Ltd. in Mumbai, India, and distributed by Ascend Laboratories, LLC in Parsippany, New Jersey. The tablets are dispensed in a USP tight container, with 60 tablets per bottle. Additional labeling may contain a UV varnish, with barcode and a serial identification number. A medical license number (MBI0S/106) and PLS code (PLS0022T) are also listed. This medication is only available with a prescription.*
evero- - evero
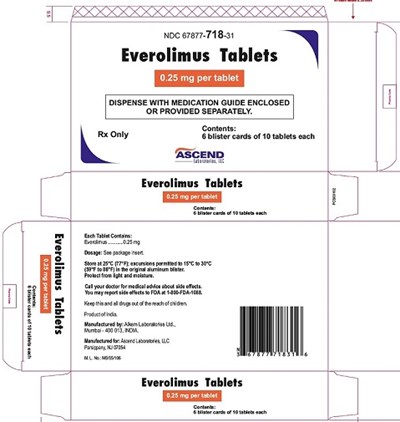
This appears to be a description of a medication called Everolimus Tablets. The tablets are dispensed with a medication guide enclosed or provided separately. The tablets come in a container with 10 tablets each, and each tablet has 0.25 mg of the medication. The medication is only available with a prescription.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.