Product Images Leucovorin Calcium
View Photos of Packaging, Labels & Appearance
- Leucovorin Calcium for Injection Cartons Rev 11 2021 Page 1
- Leucovorin Calcium for Injection Cartons Rev 11 2021 Page 2
- Leucovorin Calcium for Injection Cartons Rev 11 2021 Page 3
- Leucovorin Calcium for Injection Labels Rev 11 2021 Page 1
- Leucovorin Calcium for Injection Labels Rev 11 2021 Page 2
- Leucovorin Calcium for Injection Labels Rev 11 2021 Page 3
- Leucovorin Calcium For Injection rev 1019 - image 01
- Leucovoring Calcium for INjection 200 mg vial rev 1019 - image 02
- Leucovorin Calcium for Injection Carton 100mg Vial rev 1019 - image 03
- Leucovorin Calcium For Injection Carton 200 mg rev 10 19 - image 04
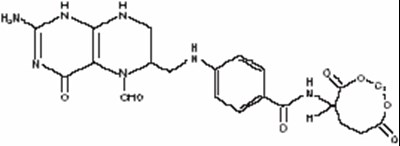
- structural formula - leucovorin calcium for injection 1
- Leucovorin Calcium for Injection 350mg Label Rev 08-19 - leucovorin calcium for injection 5
- Leucovorin Calcium for Injection 350mg Carton Rev 08-19 - leucovorin calcium for injection 9
Product Label Images
The following 13 images provide visual information about the product associated with Leucovorin Calcium NDC 68001-416 by Bluepoint Laboratories, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Leucovorin Calcium for Injection Cartons Rev 11 2021 Page 1

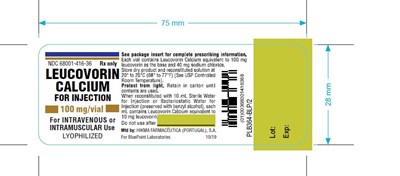
This text contains information related to a medical product called Leucovorin Calcium for Injection. Each vial of this product contains 100 mg of Leucovorin Calcium and 40 mg of Sodium Chloride. There are instructions to reconstitute the product using Bacteriostatic Water for Injection (preserved with benzyl alcohol) or Sterile Water for Injection, based on usage within 7 days or immediate use, respectively. The product is to be used for intravenous or intramuscular use only. It is manufactured by Hikma Farmaceutica in Portugal for BluePoint Laboratories in the USA, and stored at 20°C to 25°C. The packaging features a barcode, dimensions, and other information.*
Leucovorin Calcium for Injection Cartons Rev 11 2021 Page 2

Leucovorin Calcium for Injection is a medication available in vials containing 200mg of Leucovorin Calcium and 80mg of sodium chloride. It should be reconstituted with Bacteriostatic Water for Injection or Sterile Water for Injection, depending on the intended use. When diluted in 20 mL of sterile diluent, each mL of the resulting solution contains 10mg of Leucovorin Calcium. The medication is intended for intravenous or intramuscular use and should only be reconstituted with Sterile Water for Injection, USP for doses greater than 10mg/m². The product must be stored at 20°C to 25°C (68°F to 77°F), protected from light, until used. Complete prescribing information is available in the package insert.*
Leucovorin Calcium for Injection Cartons Rev 11 2021 Page 3

This is a description of Leucovorin Calcium for Injection, USP, a medication that is administered intravenously or intramuscularly. Each package contains a single-dose vial with a dry product that needs to be reconstituted with sterile or bacteriostatic water. Once reconstituted, each vial contains 350mg of Leucovorin Calcium and 140mg of sodium chloride. The medication needs to be stored at a controlled room temperature and protected from light. The package insert should be consulted for the complete prescribing information. The description also includes information about the manufacturer and the product identifier.*
Leucovorin Calcium for Injection Labels Rev 11 2021 Page 1

This appears to be a package insert for Leucovorin Calcium, containing information on dosage and storage instructions. It advises to refer to the package insert for complete prescribing information, and also notes that each vial contains 100mg of Leucovorin Calcium and 40mg of sodium chloride. It should be stored at 20° to 25°C (68° to 77°F) and protected from light until its contents are used. Once reconstituted, each mL contains 10mg of Leucovorin Calcium. The manufacturer is listed as Hikma Farmageutica (Portugal), and the expiration date and lot number are also included. No non-English characters were detected.*
Leucovorin Calcium for Injection Labels Rev 11 2021 Page 2

This is a description of a medication containing Leucovorin Calcium with a strength of 200mg per vial. It is used for intravenous or intramuscular injection and is stored in controlled room temperature. The medication must be protected from light and should be stored in a carton until contents are used. It is manufactured by Hikua Farmaceutica in Portugal and distributed by BluePoint Laboratories. Complete prescribing information can be found in the package insert. The lot number and expiration date are not listed.*
Leucovorin Calcium for Injection Labels Rev 11 2021 Page 3

This is a description of Leucovorin Calcium injection for intravenous or intramuscular use. Each vial contains 350 mg of Leucovorin Calcium and 140 mg of sodium chloride. When reconstituted, each mL contains 20 mg of Leucovorin Calcium. This medication is to be stored at 20°C to 25°C and protected from light. The manufacturer is Hikma Farmageutica and the lot number and expiration date are not available. Note that this product is for Rx only.*
Leucovoring Calcium for INjection 200 mg vial rev 1019 - image 02

The text appears to be a label of a medication named LEUCOVORIN. It comes in a lyophilized form for injection. Each vial contains Leucovorin Calcium, equivalent to 200 mg. Further details about the medication, such as usage instructions or side effects, would be available in the package insert. The rest of the text on the label seems to be information related to the ingredients and the manufacturing process, including the recommendation to store it at room temperature.*
Leucovorin Calcium for Injection Carton 100mg Vial rev 1019 - image 03

This seems to be a product description for a medication called Leucovorin, which is available as a lyophilized (freeze-dried) powder for injection. It is prescribed to patients who require doses of folate to maintain their health, especially when they are undergoing chemotherapy treatment for cancer. The text provides information on how to reconstitute the powder for injection, how to administer it, and its storage requirements. It also warns against using preservative-containing Leucovorin solutions for higher doses and advises reading the package insert for more detailed NDC (National Drug Code) numbers.*
Leucovorin Calcium For Injection Carton 200 mg rev 10 19 - image 04

This seems to be a medical document containing information on a drug called Leucovorin Calcium with instructions on dosage, storage, and reconstitution. The document also includes a warning against using the solution for doses greater than 10 mg/m² and advises to consult the package insert for complete prescribing information. The drug is available in vials containing 200mg of Leucovorin Calcium equivalent to 1010mg Leucovorin in a sterile, lyophilized form, and should be stored at controlled room temperature.*
Leucovorin Calcium for Injection 350mg Label Rev 08-19 - leucovorin calcium for injection 5

This text provides information on a product containing Leucovorin Calcium, a medication used for various purposes including treating certain types of cancer and reducing side effects of other medications. This medication comes in a dry form and requires reconstitution with sterile water. Each vial of the medication provides 350 mg of leucovorin and 140 mg of sodium chloride. When reconstituted, each milliliter of the solution contains 20 mg of leucovorin. The medication should be stored at controlled room temperature and protected from light. The text also indicates that the medication is a single-use vial and does not contain any preservatives.*
Leucovorin Calcium for Injection 350mg Carton Rev 08-19 - leucovorin calcium for injection 9

LEUCOVORIN CALCIUM FOR INJECTION is a lyophilized powder that is used for intravenous or intramuscular use. Each single-use vial contains 350 mg of leucovorin calcium equivalent to 10,350 mg of leucovorin and 140 mg of sodium chloride. The reconstituted solution should be stored at 20°C to 25°C (68°F to 77°F) and protected from light. The product does not contain any preservatives. Consult the package insert for complete prescribing information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.