Image 01

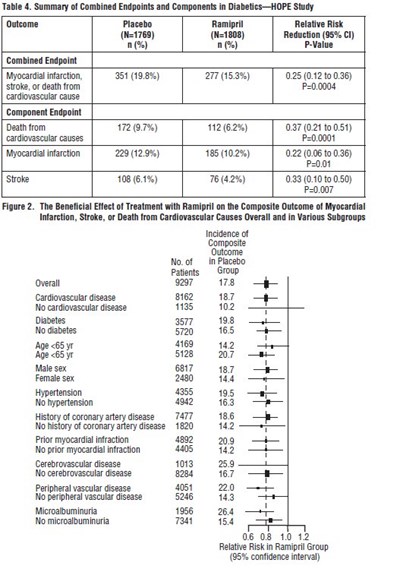
Table 3 shows the combined components and endpoints of the HOPE study, which evaluated the effects of placebo and Ramipril. The combined endpoint included myocardial infarction, stroke, or death from a cardiovascular cause. Ramipril showed a significant reduction in this endpoint compared to placebo. Component endpoints included death from cardiovascular causes, myocardial infarction, and stroke, with Ramipril showing a significant reduction in all three compared to placebo. Overall mortality was also reduced with Ramipril.*