Product Images Enoxaparin Sodium
View Photos of Packaging, Labels & Appearance
- Enoxaparin Sodium Injection 30mg/0.3mL Carton - Carton 5601A
- Enoxaparin Sodium Injection 40mg/0.4mL Carton - Carton 5602A
- enoxaparin Sodium Injection 60mg/0.6mL Carton - Carton 5603A
- Enoxaparin Sodium 80mg/0.8mL Carton - Carton 5604A
- Enoxaparin Sodium Injection 100mg/ml Carton - Carton 5605A
- Enoxaparin Sodium Injection 120mg/0.8mL Carton - Carton 5606A
- Enoxaparin Sodium Injection 150mg/mL Carton - Carton 5607A
- Enoxaparing Sodium Injection USP Vial Carton - Carton 5608
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 00
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 01
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 02
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 03
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 04
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 05
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 06
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 07
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 08
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 09
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 10
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 11
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 12
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 13
- bc9f03ca 569e 4194 ba1d 38eeee7cb56f 14
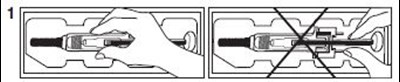
- Instructions 1 - enoxaparin 00
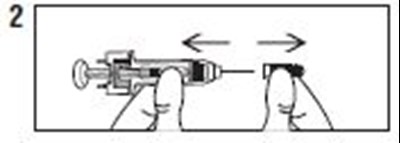
- Instructions 2 - enoxaparin 01
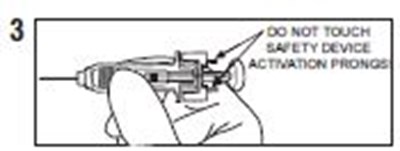
- Instructions 3 - enoxaparin 02
- Instructions 4 - enoxaparin 03
- Instructions 5 - enoxaparin 04
- Instructions 6 - enoxaparin 05
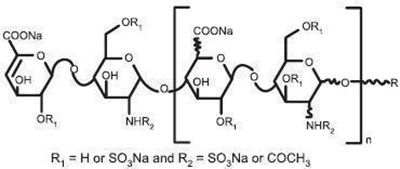
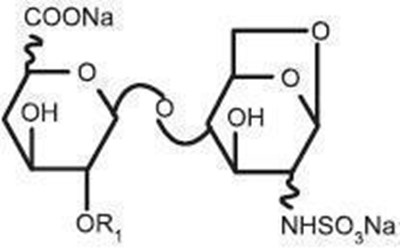
- Chemical Structure - enoxaparin 06
- Chemical Structure - enoxaparin 07
- Figure 1 - enoxaparin 08
- Figure 2 - enoxaparin 09
- Enoxaparin Sodium Injection 30mg/ 0.3mL Syringe Label - image 01
- Enoxaparin Sodium Injection 60mg/ 0.6mL - image 02
- Enoxaparing Sodium Injection 40mg/0.4mL Syringe Label - image 03
- Enoxaparin Sodium Injection 80mg/ 0.8mL Syringe Label - image 04
- Enoxaparin Sodium Injection 100mg/mL Syringe Label - image 05
- Enoxaparing Sodium Injection 120mg/ 0.8mL Syringe Label - image 06
- Enoxaparin Sodium Injection 150mg/mL Syringe Label - image 07
- Enoxaparin Sodium 300mg/3mL Vial Label - image 08
Product Label Images
The following 41 images provide visual information about the product associated with Enoxaparin Sodium NDC 68001-463 by Bluepoint Laboratories, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Enoxaparin Sodium Injection 30mg/0.3mL Carton - Carton 5601A

The text contains some alphanumeric characters and scattered letters. However, it seems to be a list of medical products. The available information mentions "ENOXAPARIN SODIUM INJECTION" and "Single Dosa Syringes." The text also shows the amount of "30 mg". The provided text seems to be an incomplete document.*
Enoxaparin Sodium Injection 40mg/0.4mL Carton - Carton 5602A

This is a description of a medication. The name of the medication is Enoxaparin Sodium Injection, which comes in a package of 10 single-dose syringes containing 40mg of the medication in each syringe. The medication is meant for subcutaneous injection only and must be stored at temperatures between 25°C and 30°C. The package contains instructions for use. The warning on the package advises to keep the medication out of the reach of children.*
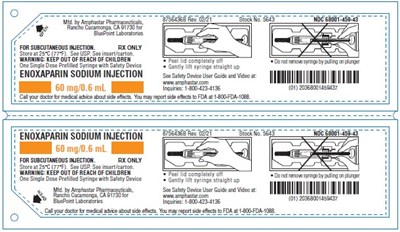
enoxaparin Sodium Injection 60mg/0.6mL Carton - Carton 5603A

This is a description of a medication called Enoxaparin Sodium Injection for subcutaneous injection. The medication comes in single dose syringes containing 60mg of the injection per 0.1ml. The injection contains 80mg of sodium and the package includes instructions for use. The medication must be stored at 25°C and not be kept out of reach of children. The patient should call their doctor or medical advisor for advice on use and side effects. There is also a warning to keep the medicine out of reach of children.*
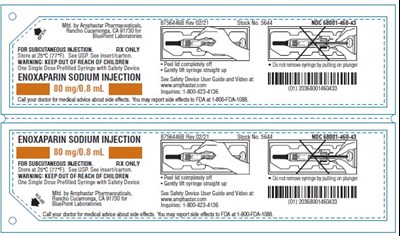
Enoxaparin Sodium 80mg/0.8mL Carton - Carton 5604A

ENOXAPARIN SODIUM INJECTION is a medication for subcutaneous injection. It is derived from porcine intestinal tissue and contains 80mg of enoxaparin sodium in a single-dose syringe. The medication is used for thrombosis prevention and treatment in patients with conditions such as deep vein thrombosis or pulmonary embolism. It is recommended to consult a doctor or medical professional before using the medication and store it at controlled room temperature. The text also includes a warning to keep the product out of reach of children.*
Enoxaparin Sodium Injection 100mg/ml Carton - Carton 5605A

ENOXAPARIN SODIUM INJECTION is a medication used for subcutaneous injection. Each milliliter contains 100 mg of noxaparin sodium derived from porcine intestinal mucosa. It is important to contact a doctor for advice on possible side effects, and it should be stored at a temperature of 2-8°C. This medication is available in single dose syringes of 25 mg. The text includes a warning to keep the medication out of the reach of children.*
Enoxaparin Sodium Injection 120mg/0.8mL Carton - Carton 5606A

This description is for a medication called Enoxaparin Sodium Injection. It comes in single-dose syringes containing 120mg of the medication, which is derived from porcine intestinal mucosa. The medication is meant for subcutaneous injection and the package insert should be consulted for instructions on the proper use and administration. The medication should be stored at 25°C with permitted excursions.*
Enoxaparin Sodium Injection 150mg/mL Carton - Carton 5607A

This is a description of a medication packaging for Enoxaparin Sodium Injection, used for subcutaneous injections. The package contains 10 single-dose syringes, with each 1 mL dosage measuring 150 mg of Enoxaparin Sodium. It provides storage instructions and cautions to keep it out of reach of children. The text also mentions a reference number and contact information to call for medical advice or in case of adverse effects.*
Enoxaparing Sodium Injection USP Vial Carton - Carton 5608

This is a description of a pharmaceutical product called Enoxaparin Sodium. It is an injection used for subcutaneous or intravenous administration. The solution contains 300 mg/3mL or 100 mg/mL of enoxaparin sodium, which is derived from porcine intestinal mucosa. The package insert should be consulted for dosage information and directions for use. The multiple-dose vial should not be stored for more than 28 days after the first use and should be kept away from children. The product is manufactured by Amphastar Pharmaceuticals in Rancho Cucamonga, California.*
bc9f03ca 569e 4194 ba1d 38eeee7cb56f 00
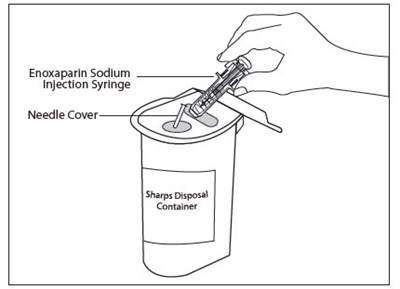
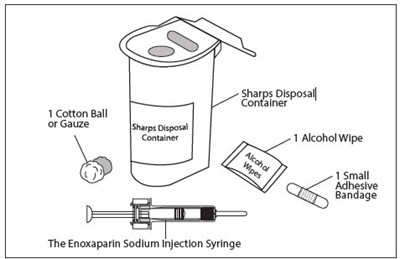
This text seems to be describing a medical product called "Enoxaparin Sodium Injection Syringe" which comes with a needle cover. There is also a sharp disposal container included for safe disposal of the used syringe and needle.*
bc9f03ca 569e 4194 ba1d 38eeee7cb56f 01

This text is already a description of the item "Protective Sleeve", which means it does not need any further description.*
bc9f03ca 569e 4194 ba1d 38eeee7cb56f 10

Clear Liquid is a term used to describe a liquid substance that is transparent, without any visible particles or impurities. This type of liquid can include purified water, certain types of juices, broths, and some medications. It is commonly used in medical settings as a preparation for diagnostic tests, surgeries, or as a part of a cleansing process. Clear liquid diets are also recommended for those recovering from certain medical conditions or preparing for medical procedures.*
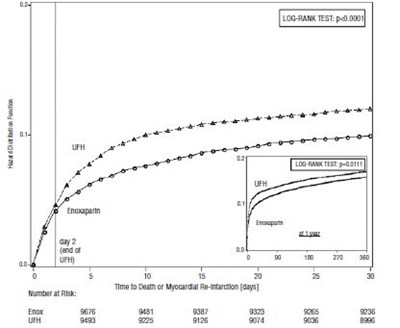
Figure 1 - enoxaparin 08

This is a table containing information about a medical study. The study gathered data on subgroups of patients separated by sex, age, infarct location, diabetes, prior MI, fibrinolytic agent, and time to treatment. The table shows the number of patients in each subgroup, the number of cardiac events (UFI), and the reduction in the risk of cardiac events with UFH treatment. The last row indicates that UFH (unfractionated heparin) is better than the alternative treatment, but it's unclear what the alternative treatment was.*
Enoxaparing Sodium Injection 40mg/0.4mL Syringe Label - image 03

This is a description of a medication named Enoxaparin Sodium Injection by Amphastar Pharmaceuticals. The injection is meant for subcutaneous use only, and each 0.4mL contains 40mg of the medication. There is a warning to keep the injection out of reach of children and caution to use only a proper syringe. The rest of the text is not readable.*
Enoxaparin Sodium Injection 100mg/mL Syringe Label - image 05

This appears to be a mix of random letters, symbols, and phrases that do not form a coherent description. Therefore, it is not available.*
Enoxaparin Sodium 300mg/3mL Vial Label - image 08

Enoxaparin Sodium is a prescription drug available as an injection in a 3 mL vial with a concentration of 100 mg/mL. The medication is intended for subcutaneous or intravenous use and must be stored at 20°C to 25°C. It is recommended not to use the same vial for more than 28 days. The drug is manufactured by Hastar Pharmaceuticals Inc. for BluePoint Laboratories.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.