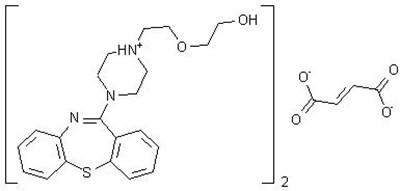
Product Images Quetiapine Fumarate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Quetiapine Fumarate NDC 68001-513 by Bluepoint Laboratories, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Quetiapine Extended-Release Tablets 200mg (NDC 68001-513-06) - quetiapine 200mg 60 label

This is a medication called Quetiapine, with each tablet containing 230mg of Quetiapine Fumarate equivalent to Quetiapine 200mg. The tablets must be stored at a temperature of 20°C to 25°C, and must be swallowed whole, without splitting, crushing or chewing. The usual dosage is once daily, and a warning is given to keep medication out of children’s reach. The medication is manufactured by Intas Pharmaceuticals Limited in India, and is for BluePoint Laboratories. The packaging contains 60 tablets, and the prescription must be dispensed along with the enclosed medication guide. There is a NDC (National Drug Code) number and other identifying information related to the manufacturer.*
Quetiapine Extended-Release Tablets 300mg (NDC 68001-514-06) - quetiapine 300mg 60 label

This is a medication called Quetiapine, specifically its extended-release variant, with each tablet containing 345mg of Quetiapine Fumarate, equivalent to 300mg of Quetiapine. The tablets should be stored between 20°C to 25°C and should not be split, crushed or chewed as it must be swallowed whole. The usual dosage is once a day, as documented in the accompanying prescribing information. It is manufactured by Intas Pharmaceuticals Limited in Ahmedabad, India, and distributed by BluePoint Laboratories under the NDC code 68001-514-06. The enclosed medication guide should be dispensed to each patient by a pharmacist.*
Quetiapine Extended-Release Tablets 400mg (NDC 68001-515-06) - quetiapine 400mg 60 label

Each tablet of Quetiapine Extended-Release contains 461 mg of Quetiapine Fumarate, USP that is equal to Quetiapine 400 mg. This medication should be stored between 20°C to 25°C (68°F to 77°F) and should not be split, crushed or chewed. The recommended dosage is once daily as indicated in the accompanying prescribing information. This medication should be kept out of the reach of children and is manufactured by Intas Pharmaceuticals Limited in Ahmedabad, India. The product is meant for prescription-only use, and BluePoint Laboratories is the manufacturer with a manufacturing license number of G/25/1339. The package also includes a medication guide that the pharmacist should provide to each patient.*
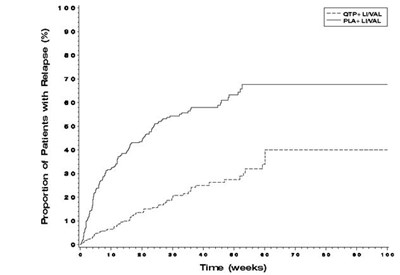
Figure 3 Kaplan-Meier Curves of Time to Recurrence of a Mood Event (Study 19).jpg - quetiapine figure 3

This appears to be a graph presenting the proportion of patients who experienced a relapse over time in weeks. The proportion starts at 100% and there are two lines labeled "VAL" and "PLac LwAL". However, without additional context or data it is unclear what the values represent.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.