Product Images Pemetrexed
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Pemetrexed NDC 68001-539 by Bluepoint Laboratories, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - pemetrexed 03

This table provides information on the survival probability for patients treated with either Pemetrexed + Cisplatin (PC) or Gemcitabine + Cisplatin (GC). The table shows survival time in months and the number of patients at risk for different time intervals.*
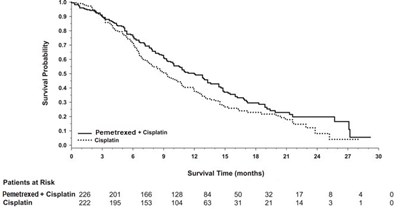
Figure 3 - pemetrexed 04

This is a survival probability graph comparing two chemotherapy treatments for patients. The first treatment is labeled "Pemetrexed + Cisplatin" and abbreviated as "PC" while the second treatment is labeled "Gomcitabine + Cisplatin" and abbreviated as "GC". The X-axis represents the survival time in months while the Y-axis represents the probability of survival. The graph shows that PC has a higher survival probability than GC for the first 6 months, after which the survival probability of GC surpasses that of PC. The number of patients at risk is also represented on the graph for each treatment at various times.*
Figure 4 - pemetrexed 05

This is a survival probability chart representing the survival probability for two drug combinations; Pemetrexed + Cisplatin (PC) and Gemcitabine + Cisplatin (GC). The chart shows the survival time in months and patients at risk. However, there is not enough information to interpret the chart to offer further insight.*
Figure 5 - pemetrexed 06

The text is describing a survival probability chart for patients who were given Pemetrexed and Placebo. It shows the number of patients at risk and their respective survival rates in months. There is not enough context to determine the context of the study or the outcome for patients.*
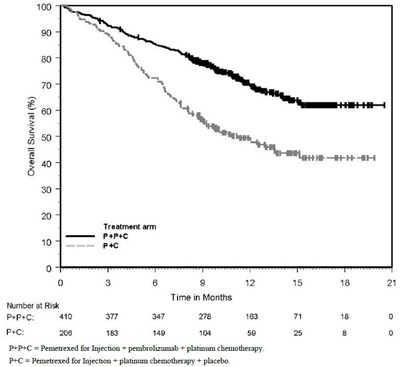
Figure 6 - pemetrexed 07

This appears to be a survival analysis chart displaying the survival probability of patients at risk for a treatment group (Pemetrexed) and a control group (Placebo) over a certain period of time (in months). The number of patients at risk in each group is provided for each time point. However, some characters such as the quotation mark and the letter "e" are not clear, making it difficult to interpret the exact values.*
Figure 7 - pemetrexed 08

This is a table that displays the survival probability of patients receiving Pemetrexed and Placebo, along with the number of patients at risk for each treatment. The table also shows the survival time of the patients in months.*
Figure 8 - pemetrexed 09

This is a survival probability table showing the number of patients at risk who received Pemetrexed and Placebo. The table also includes the survival time in months.*
Figure 9 - pemetrexed 10
The text appears to be a table showing the survival time (in months) of patients at risk who received treatment with Pemetrexed+Cisplatin.*
Vial Label-Pemetrexed for Injection 100 mg Single-dose Vial - pemetrexed 11

Each vial contains pemetrexed disodium equivalent to 100mg pemetrexed, 106mg of mannitol. Hydrochloric acid and/or sodium hydroxide may have been added to adjust pH. Pemetrexed storage should be at 25°C; permitted excursions for injection, USP is 15°C to 30°C. It's a Controlled Room Temperature storage drug. There is accompanying literature for storage of reconstituted and infusion solutions. It is to be used intravenously, with caution as it is a Cytotoxic Agent. The text also mentions the manufacturer, BluePoint Laboratories, and a batch number L¥8¢6,1 0089.*
PACKAGE CARTON- Pemetrexed for Injection 100 mg single-dose Vial - pemetrexed 12

This is a description of a drug called Pemetrexed, provided in a vial containing 100 mg of the medication. The vial also contains 106 mg of Mannitol. The text explains that Hydrochloric acid and/or sodium may have been added to adjust pH. The drug is meant for intravenous use only and is a cytotoxic agent. The accompanying literature should be referred to for the correct dosage. To reconstitute the drug, 4.2 mL of 0.8% Sodium Chloride Injection should be added to the vial. This will create a solution with 25 mo/mL of Pemetrexed. The reconstituted solution should be further diluted before use. Once reconstituted, the solution should be kept at a refrigerated temperature of 2°C to 8°C (35°F to 46°F). The drug should be discarded if not used within 24 hours of reconstitution. Finally, the drug should be stored at a temperature of 25°C (77°F), with excursions permitted between 15°C to 50°C (59°F to 86°F). The manufacturer of the drug is Qilu Pharmaceutical.*
VIAL LABEL: Pemetrexed for Injection 500 mg single-dose vial - pemetrexed 14

Each vial contains pemetrexed disodium equivalent to 500mg pemetrexed and 500mg of mannitol. Hydrochloric acid and/or sodium hydroxide may have been added to adjust pH. The powder should be stored at 25°C and excursions between 15°C and 30°C is allowed for injection. The storage instructions for reconstituted and infusion solutions can be found in the accompanying literature. It is a cytotoxic agent for intravenous use only. The product is manufactured by Qilu Pharmaceutical (Hainan) Co., Ltd for BluePoint Laboratories.*
PACKAGE CARTON: Pemetrexed for Injection 500 mg single dose vial - pemetrexed 15

This is a description of a pharmaceutical product called Pemetrexed, designed for intravenous use as a cytotoxic agent. The product comes in a single-dose vial and contains 500mg of Pemetrexed disodium and 500mg of mannitol. The pH is adjusted using hydrochloric acid and/or sodium hydroxide. To reconstitute, one needs to add 20 mL of 0.9% Sodium Chloride Injection. The reconstituted solution should be further diluted before use, and infusion should be administered within 24 hours after initial reconstitution. The accompanying literature provides information about the dosage required; storage for the powder is at 25°C while reconstituted and infusion solutions are refrigerated at 2°C to 8°C.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.