Product Images Pemetrexed
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 12 images provide visual information about the product associated with Pemetrexed NDC 68001-545 by Bluepoint Laboratories, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
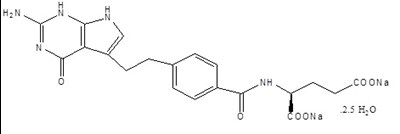
Pemetrexed for Injection 750mg Label - image 01

This text is a description of a drug called Pemetrexed, which is for Injection and only to be used intravenously. It comes in a single-dose vial called OncoPoint and each vial contains pemetrexed disodium equivalent to 750 mg pemetrexed, 750 mg of mannitol. It is a Cytotoxic Agent and caution should be taken while handling it. The storage temperature is between 20°C to 25°C (68 to 77°F), and the literature accompanying the drug should be consulted for the storage of reconstituted and infusion solutions. It is manufactured by lnd Phama L, Ind. for BusPont Laboratories.*
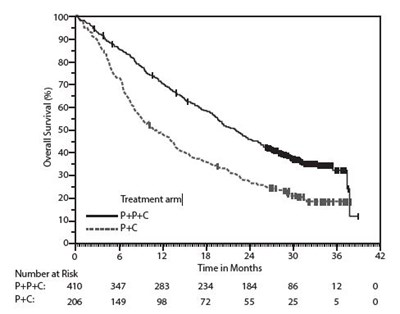
Figure 2 - image 05

This looks like a data table showing survival probabilities and time in months. There are two columns, one titled "Survival Probability" and one titled "Survival Time." The data in the table appears to be numerical values, but without further context or information it's difficult to interpret their meaning.*
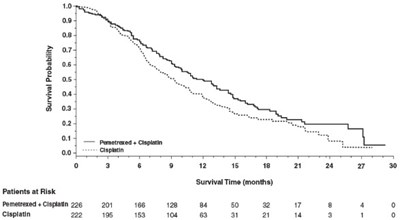
Figure 3 - image 06

This appears to be medical data, likely related to a study or research on a drug combination called Gometabin + Clpi. The rest of the text includes patient IDs and survival time, measured in months. There is no other useful information available.*
Figure 5 - image 08
This appears to be a graph or chart with data related to "Survival Probability" and "Survival Time (months)". The X-axis shows the time in months, while the Y-axis shows the survival probability. The chart may have additional features or content, but without further context, this is the only information that can be determined from the provided text.*
Figure 6 - image 09

This appears to be a graph showing survival probability for two groups of patients - one who received "Pometocea" and another who received a "Placebo." The x-axis appears to show survival time in months, while the y-axis shows the proportion of patients who survived up to that time. There are also numbers indicating the number of patients at risk in each group at different time points. No further information can be discerned from the text provided.*
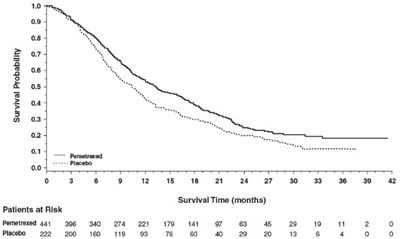
Figure 8 - image 11

This is a medical report of two groups of patients, one treated with Pematrexed (with 350 patients at the beginning) and the other treated with Placsbo (with 180 patients at the beginning). The report includes the number of patients at risk after a certain period of time (indicated by the numbers at the bottom). There is not enough information to determine the time interval or what "aw" and "2" represent.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.