Product Images Mitomycin
View Photos of Packaging, Labels & Appearance
- Label Mitomycin for injection USP 5 mg per vial - mitomycin fig1
- Carton Mitomycin for injection 5 mg USP per vial - mitomycin fig2
- Label Mitomycin for injection USP 20 mg per vial - mitomycin fig3
- Carton Mitomycin for Injection USP 20 mg per vial - mitomycin fig4
- Label Mitomycin for Injection USP 40 mg per vial - mitomycin fig5
- Carton Mitomycin for Injection USP 40 mg per vial - mitomycin fig6
- mitomycin-str - mitomycin str
Product Label Images
The following 7 images provide visual information about the product associated with Mitomycin NDC 68001-617 by Bluepoint Laboratories, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Label Mitomycin for injection USP 5 mg per vial - mitomycin fig1

This is a description of a vial containing mitomycin, USP 5 mg, and mannitol 10 g. The medication is for injection and must be stored in the container within the carton, at temperatures between 15°C and 30°C, protected from light. The reconstituted solution needs to be stored under refrigeration at 2°C to 8°C and should be discarded after 14 days. The vial is a single-dose vial and needs to be used within 7 days after reconstitution. The product is manufactured by Eugia Pharma Specialties Limited. Administering this medication intravenously is essential.*
Carton Mitomycin for injection 5 mg USP per vial - mitomycin fig2
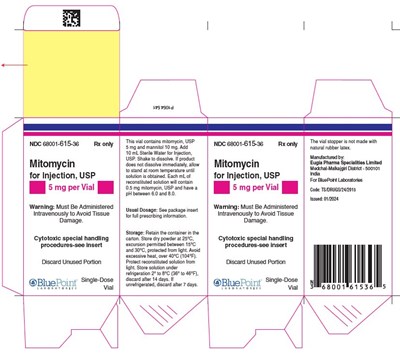
This is a pharmaceutical product description for Mitomycin for Injection, USP. It is a cytotoxic drug that must be administered intravenously to avoid tissue damage. The drug comes in vials containing 5mg of Mitomycin and 10mg of mannitol, which needs to be dissolved in 10mL of Sterile Water for Injection, USP. The usual dosage and storage instructions are provided on the packaging. The vials are single-dose and come with specific handling and disposal guidelines. The manufacturing information is also included in the description.*
Label Mitomycin for injection USP 20 mg per vial - mitomycin fig3

This text provides detailed instructions for the storage and administration of a medication called mitomycin, which comes in a dry powder form to be reconstituted for injection. The storage temperature range, reconstitution process, dosage information, and warnings are all clearly outlined. It emphasizes the importance of proper storage conditions, avoiding light exposure, and timely discarding of any unused portions.*
Carton Mitomycin for Injection USP 20 mg per vial - mitomycin fig4

This text appears to be a description of a medication called Mitomycin for Injection, USP, which is used for certain medical treatments. It contains specific instructions on how to administer the drug intravenously, handle it properly, and store it safely. The medication comes in 20 mg per vial and needs to be mixed with sterile water for injection before use. It also includes details on the dosage, storage conditions, and shelf life after reconstitution. Additionally, the text mentions the manufacturer of the drug and provides a warning about tissue damage if not administered correctly.*
Label Mitomycin for Injection USP 40 mg per vial - mitomycin fig5

Mitomycin for Injection, USP is a medication administered intravenously to prevent tissue damage. Each vial contains 40mg of mitomycin USP and 80mg of mannitol. The product should be dissolved in Sterile Water for Injection and is a single-dose vial. After reconstitution, the solution should be stored under refrigeration and discarded after 14 days. It is important to follow the package insert for full prescribing information and storage instructions to ensure efficacy and safety. This product is manufactured in India by Eugia Pharma Specialities Limited for BluePoint Laboratories.*
Carton Mitomycin for Injection USP 40 mg per vial - mitomycin fig6

This text provides information about Mitomycin for Injection, USP. It includes details on dosage, administration warnings, storage instructions, and manufacturing information. Mitomycin is a cytotoxic drug that must be administered intravenously to avoid tissue damage. It is important to follow specific handling procedures, discard any unused portion, and store the solution properly. The text also mentions the contents of the vial and the process of reconstitution. Furthermore, it highlights the importance of proper storage temperatures and the expiration dates for both the dry powder and reconstituted solution. The manufacturer mentioned in the text is Eugia Pharma Specialities Limited located in India.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
