Product Images Enoxaparin Sodium
View Photos of Packaging, Labels & Appearance
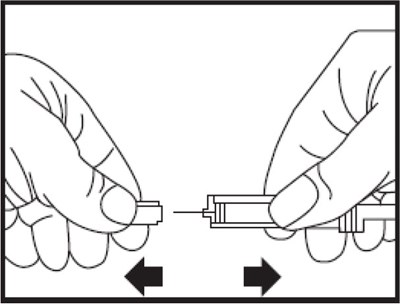
- Figure A - eno0f 0001 01
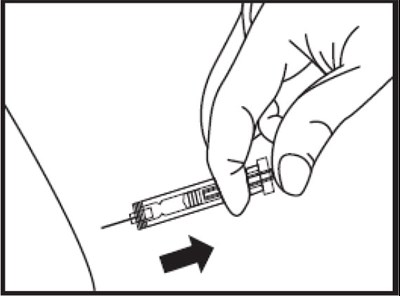
- Figure B - eno0f 0001 02
- Figure C - eno0f 0001 03
- Figure D - eno0f 0001 04
- Figure E - eno0f 0001 05
- figure - eno0f 0001 06
- figure - eno0f 0001 07
- figure 1 - eno0f 0001 08
- figure 2 - eno0f 0001 09
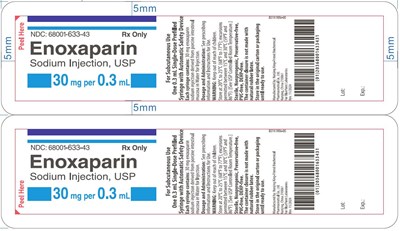
- Label- Enoxaparin Sodium Injection USP 30 mg - eno0f 0001 10 v1
- Carton- Enoxaparin Sodium Injection USP 30 mg - eno0f 0001 11 v2
- Label- Enoxaparin Sodium Injection USP 40 mg - eno0f 0001 12 v1
- Carton- Enoxaparin Sodium Injection USP 40 mg - eno0f 0001 13 v2
- Label- Enoxaparin Sodium Injection USP 60 mg - eno0f 0001 14 v1
- Carton- Enoxaparin Sodium Injection USP 60 mg - eno0f 0001 15 v2
- Label- Enoxaparin Sodium Injection USP 80 mg - eno0f 0001 16 v1
- Carton- Enoxaparin Sodium Injection USP 80 mg - eno0f 0001 17 v2
- Label- Enoxaparin Sodium Injection USP 100 mg - eno0f 0001 18 v1
- Carton- Enoxaparin Sodium Injection USP 100 mg - eno0f 0001 19 v2
- Label- Enoxaparin Sodium Injection USP 120 mg - eno0f 0001 20 v1
- Carton- Enoxaparin Sodium Injection USP 120 mg - eno0f 0001 21 v2
- Label- Enoxaparin Sodium Injection USP 150 mg - eno0f 0001 22 v1
- Carton- Enoxaparin Sodium Injection USP 150 mg - eno0f 0001 23 v2
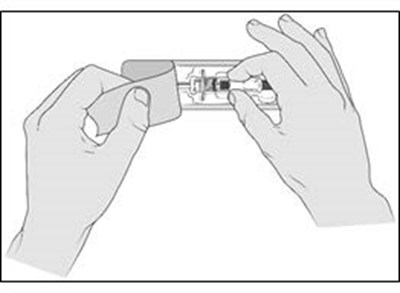
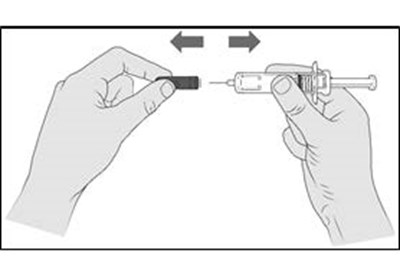
- Figure A - enox ifu a
- Figure B - enox ifu b
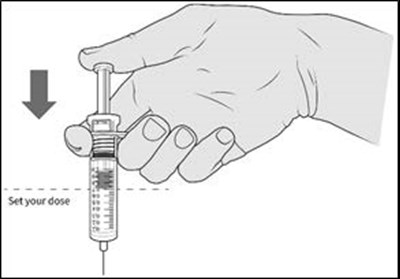
- Figure C - enox ifu c
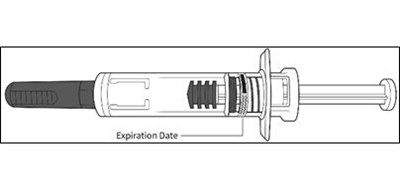
- Figure D - enox ifu d
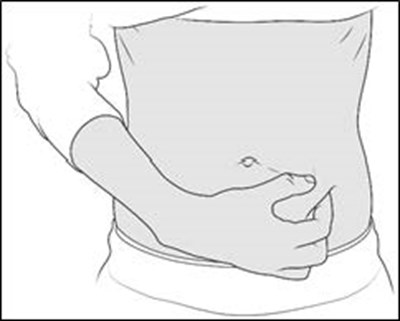
- Figure E - enox ifu e
- Figure F - enox ifu f
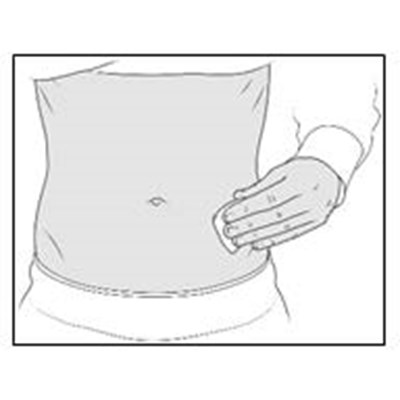
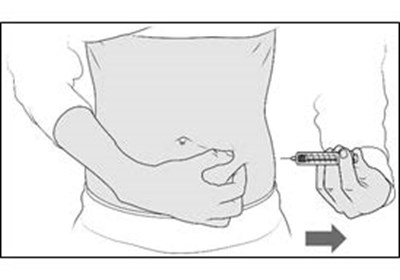
- Figure G - enox ifu g
- Figure H - enox ifu h
- Figure I - enox ifu i
- Figure J - enox ifu j
- Figure K - enox ifu k
- Figure L - enox ifu l
- Figure M - enox ifu m
- Figure N - enox ifu n
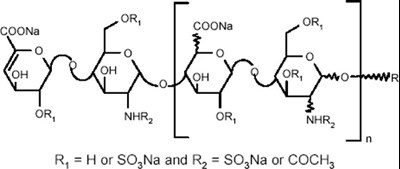
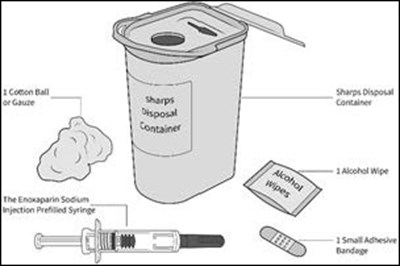
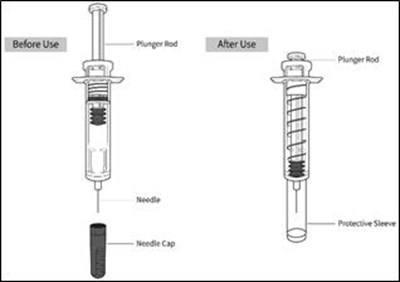
- Parts - enox ifu parts
Product Label Images
The following 38 images provide visual information about the product associated with Enoxaparin Sodium NDC 68001-634 by Bluepoint Laboratories, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure 1 - eno0f 0001 08

This text appears to provide a breakdown of a study or clinical trial subgrouped by different variables such as sex, age, infarct location, diabetes status, prior MI, type of fibrinolytic agent used, time to treatment, and the occurrence of PCl in 30 days. It also mentions the overall statistics and relative risk values. The data seems to compare the outcomes between different subgroups in terms of percentage reductions with specific treatments like Enoxaparin and UFH. This information may be valuable for medical professionals and researchers analyzing the efficacy of different treatments for specific patient groups in the context of cardiovascular events such as myocardial infarction.*
figure 2 - eno0f 0001 09
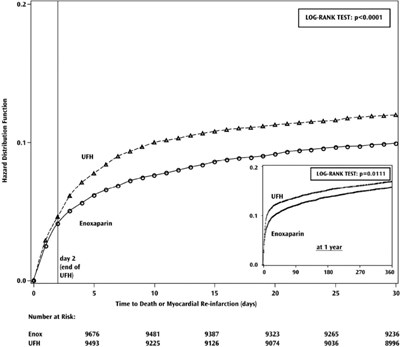
This is a hazard distribution function comparing Enoxaparin and UFH over a period of 1 year. The results of the Log-Rank Test for Enoxaparin show a p-value of less than 0.0001, indicating a significant difference. For UFH, the Log-Rank Test resulted in a p-value of 0.0111. The graph shows the time to death or myocardial re-infarction over different days, displaying the number at risk for both Enoxaparin and UFH at various time points.*
Carton- Enoxaparin Sodium Injection USP 40 mg - eno0f 0001 13 v2

This text contains information about Enoxaparin Sodium Injection, USP, including dosage, administration instructions, storage guidelines, and manufacturer details. The medication is derived from porcine intestinal mucosa and is intended for subcutaneous use. Each syringe contains 40 mg of enoxaparin sodium. Proper storage at room temperature is recommended. Manufacturer details indicate it is produced by Narjog King Fera.*
Carton- Enoxaparin Sodium Injection USP 60 mg - eno0f 0001 15 v2
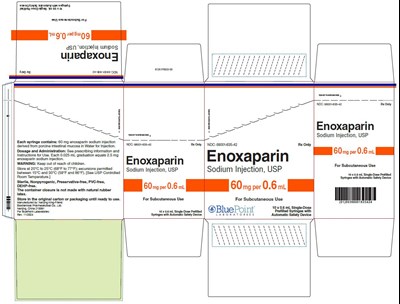
This is a description for Enoxaparin Sodium Injection, USP, used for subcutaneous administration. Each syringe contains 60 mg of enoxaparin sodium derived from porcine intestinal mucosa. The dosage and administration details can be found in the prescribing information. The product is stored between 20°C to 25°C with excursions permitted between 15°C and 30°C. The container closure is not made with natural rubber latex. Each 0.025 mL graduation equals 2.5 mg of enoxaparin sodium injection. The product is sterile, non-pyrogenic, preservative-free, PVC-free, and DEHP-free. It is recommended to store the product in the original carton until ready to use. The syringes are single-dose prefilled syringes with auto-safety syringe systems.*
Carton- Enoxaparin Sodium Injection USP 80 mg - eno0f 0001 17 v2

This is information about Enoxaparin Sodium Injection, a medication used for subcutaneous administration. The text includes details about the concentration of the injection (80mg per 0.8mL), the packaging information, and the National Drug Code (NDC) for identification purposes. It is mentioned that the medication is for subcutaneous use only. The text also indicates that the product should be stored at room temperature.*
Carton- Enoxaparin Sodium Injection USP 120 mg - eno0f 0001 21 v2

This text appears to be a description related to a medical product. It mentions details about Enoxaparin Sodium Injection, USP, its composition, dosage, administration, and storage instructions. The product is derived from porcine intestinal mucosa and is for subcutaneous use. It also includes a warning to keep the product out of reach of children. Storage recommendations are to store at 20°C to 25°C.*
Label- Enoxaparin Sodium Injection USP 150 mg - eno0f 0001 22 v1

This text appears to be a label or description for Enoxaparin Sodium Injection, USP. The product is a medication administered in milligrams per milliliter with specific instructions for use. It includes information such as the dosage strength, NDC code, and directions for use. This medication may be used for anticoagulation therapy to prevent blood clots.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.