FDA Label for Senna
View Indications, Usage & Precautions
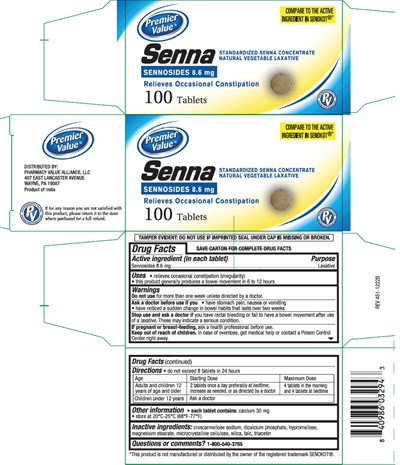
Senna Product Label
The following document was submitted to the FDA by the labeler of this product Pharmacy Value Alliance Llc. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient (In Each Tablet)
Sennosides 8.6 mg
Purpose
Laxative
Uses
- relieves occasional constipation (irregularity)
- generally produces bowel movement in 6 to 12 hours
Warnings
Do not use
for more than one week unless directed by a doctor.
Ask a doctor before use if you
• have stomach pain, nausea or vomiting
• have noticed a sudden change in bowel habits that lasts over 2 weeks
Stop use and ask a doctor if
you have rectal bleeding or fail to have a bowel movement after use of a laxative.
These may indicate a serious condition.
If pregnant or breast-feeding, ask a health professional before use.
Otc - Keep Out Of Reach Of Children
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
• do not exceed 8 tablets in 24 hours
| Age
| Starting Dose
| Maximum Dose
|
|---|---|---|
| adults and children 12 years of age and older
| 2 tablets once a day preferably at bedtime; increase as needed, or as directed by a doctor
| 4 tablets in the morning and 4 tablets at bedtime
|
| children under 12 years
| ask a doctor
|
Other Information
- each tablet contains: calcium 30 mg
- Tamper Evident: Do not use if imprinted seal under cap is missing or broken
- store at 20°C-25°C (68°F-77°F)
Inactive Ingredients
croscarmellose sodium, dicalcium phosphate, hypromellose, magnesium stearate, microcrystalline cellulose, silica, talc, triacetin
Package Label
* Please review the disclaimer below.