Product Images Sernivo
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Sernivo NDC 68040-714 by Primus Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
120 mL Carton Label - sernivo 06
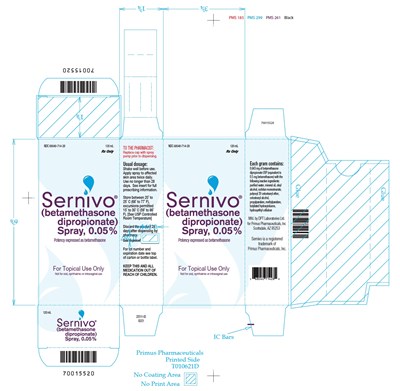
This is a medication with the NDC code 6B040-714-28. It is a spray that contains 0.05% of betamethasone dipropionate. The potency of the medication is expressed as betamethasone. The medication is for topical use only and should not be used orally, ophthalmically, or intravaginally. The medication should be shaken well before use and applied to the skin twice daily. The medication should not be used for longer than two days. The medication is manufactured by PMS 299 PMS 261 Black and the expiration date can be found on the top of the carton or bottle label. The medication comes in a spray bottle and each gram contains 0.843 mg of betamethasone dipropionate.*
120 mL Spray Bottle Label - sernivo 07

Sernivo is a topical spray medication containing 0.05% betamethasone dipropionate which is for external use only to be applied twice daily on the affected skin after shaking well. The product should be discarded after 28 days of dispensation by the pharmacy. The medication should be kept out of children's reach and stored between 20°C to 25°C. Sernivo is manufactured by DPT Laboratories Ltd. and distributed by Primus Pharmaceuticals, Inc. Scottsdale AZ 85253.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.