Product Images Tadalafil
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Tadalafil NDC 68071-2554 by Nucare Pharmaceuticals,inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
pdp - 68071 2554 5

This is a description of a medication called Tadalafil 10mg manufactured by PharmaCARE Pharmaceuticals, Inc. The medication comes in a bottle of 15 oval-shaped light yellow tablets that contain Tadalafil, USP 10mg. Each tablet is film-coated and has a debossed mark. The NDC number for the medication is 68071-2554-5, and the lot number is 00000. The medication also has an MFR NDC number of 27241-113-05, and it expires on 00-00. The product is packaged with FDA labeling and has a warning to keep it away from children and store it at a controlled temperature of 68-77°F. The packaging also includes contact information for medical advice and a report on side effects.*
figure8 - tadalafil fig8
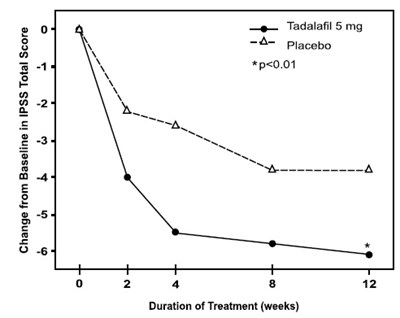
The text describes the IPSS Total Score and the change from baseline in relation to treatment with Tadalafil 5mg versus Placebo. It also indicates a statistically significant difference between the two treatments with a p-value of 0.01. However, there is no information on the actual scale of the IPSS score, the condition being treated, or any other relevant clinical details.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.