Product Images Tramadol Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Tramadol Hydrochloride NDC 68071-2945 by Nucare Pharmaceuticals,inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
pdp - 68071 2945 2

The text describes a medication manufactured by uCare Pharmaceuticals, with the NDC code 68071-29452. It is a white or off-white, capsule-shaped tablet with the inscription "ST" on one side, and the numbers 1, 4, and 5 and the letters "0" and "3" separated by a score on the other side. Each tablet contains 50 mg of Tramadol Hydrochloride USP, which may be habit-forming, so it must not be transferred to someone else. The product should be stored at a controlled temperature of 68.77°F and kept out of reach of children.*
Clinical Studies Section - tramadol fig2
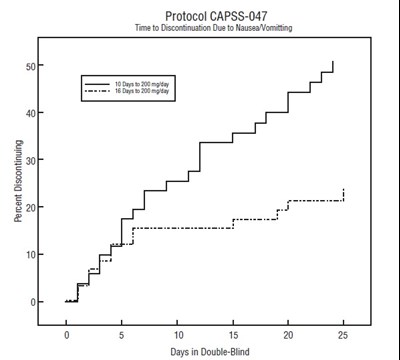
This data represents the percent of participants discontinuing the CAPSS-047 Protocol due to nausea/vomiting over a period of 20 days. The chart also shows the specific time frame of 10 days to 200 mgldey.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

