Product Images Montelukast Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Montelukast Sodium NDC 68071-4047 by Nucare Pharmaceuticals,inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
68071 4047 3

NuCare Pharmaceuticals, Inc. provides Montelukast Sodium tablets in 10mg strengths, available in 30-count bottles. Each bottle has Lot # 000000 and NDC code 68071-404/-3. The medication is also available in capsule form packed in aluminum blister packs. Each pack has NDC code Sa0y1-4047-03, and Lot # 000000, and a shelf life of Exp./=00:00. Additionally, the blister packs have a bar code (GTIN) of 0036807140473% to facilitate inventory control. However, the complete list of ingredients is only available on the manufacturer's label with the medication. To report the side effects of the medication, contact FDA or call a healthcare provider. Also, store the medication at a controlled temperature of 59-86°F and away from children.*
image of figure 1 - montelukast fig1
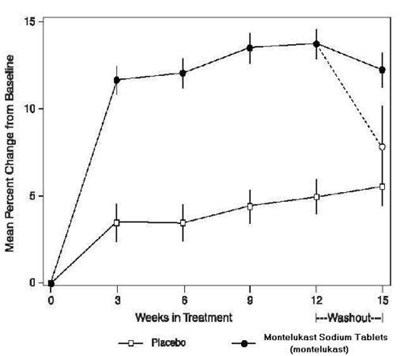
This appears to be a chart or graph showing the mean percent change from baseline for a drug called Montelukast Sodium Tablets over a period of 15 weeks in treatment. There are also references to a "F-Washout" and some numerical values but without further context, it is difficult to provide a more specific description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
