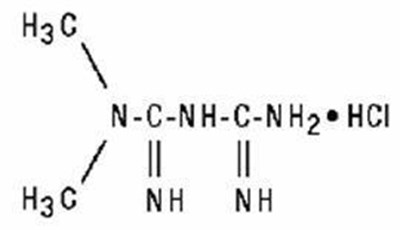
Product Images Metformin Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Metformin Hydrochloride NDC 68071-4525 by Nucare Pharmaceuticals,inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
bd3a3e6-1b64-4bda-88db-ae433d17dc8a - 0bd3a3e6 1b64 4bda 88db ae433d17dc8a

The text presents a table summarizing changes in major lipid variables after 24 weeks of treatment with Metformin Hydrochloride Tablets. The variables analyzed were: total cholesterol, total triglycerides, LDL-cholesterol, and HDL-cholesterol. The changes in percentage from baseline of each variable were measured at the final visit. However, there is not enough information to make an assessment of the efficacy of the treatment or the clinical relevance of the results.*
705cc8a-5055-412c-825a-80a3fd5b9c7a - 3705cc8a 5055 412c 825a 80a3fd5b9c7a

The text describes a study with Metformin Hydrochloride Extended Release Tablets in three doses for 24 weeks. The study evaluated the changes in Hemoglobin Ay, Fasting Plasma Glucose, and Body Weight in patients with diabetes. The changes from the baseline were recorded at week 12 and at the final visit. The study included 66 to 72 participants in each group for each measurement, and all patients received 500 mg of Metformin Hydrochloride tablets twice daily at the beginning.*
0403393-33b1-484a-a4d0-8c0a182f0dbd - 60403393 33b1 484a a4d0 8c0a182f0dbd

The text is a table showing the summary of mean percent change from baseline of major serum lipid variables at the final visit, for 29-week studies comparing Metformin Hydrochloride tablets to placebo and combined Metformin Hydrochloride tablets/Glyburide to monotherapy. The variables measured include total cholesterol, total triglycerides, LDL-cholesterol, and HDL-cholesterol, and the mean percent change for each variable is reported for each treatment type.*
68071 4525 1

This appears to be the label of a medication bottle manufactured by NuCare Pharmaceuticals, Inc. The medication is Metformin HCI, a common drug used to treat Type 2 Diabetes. The bottle contains 100 tablets of 1000mg Metformin HCI. The lot number is 000000 and the NDC number is 68071-4525-01. The manufacturer's NDC code is 23155-104-01 and the expiration date is listed as 00-00. The Global Trade Item Number (GTIN) is 0036807452518. The label contains a warning to keep the medication out of the reach of children and to store it at a controlled temperature between 59-86°F.*
dadc8a7-4fe8-4830-9bd5-82f3a0e5184c - 6dadc8a7 4fe8 4830 9bd5 82f3a0e5184c
This text provides information about a specific group of patients. The patients in question are pediatric and have a mean age of 13.8 years, with a range from 10 to 16 years old. All of the patients are on diet therapy at the beginning of the study. However, the text also notes that there is no statistically significant difference between the patients at the start of the study.*
1c10ce2-2e46-47dd-b7e7-1083fe41137b - 71c10ce2 2e46 47dd b7e7 1083fe41137b

This is a summary of changes in fasting plasma glucose, HbA, and Body Weight from baseline in a 29-week study comparing Metformin Hydrochloride Tablets to a placebo. The table shows mean changes in FPG, Hemoglobin A, and Body Weight at the final visit. The data shows that Metformin Hydrochloride Tablets produced a significant decrease in FPG and Hemoglobin A compared to the placebo. However, there was no significant difference in Body Weight between the two groups.*
48c0b5f-a8ef-49a1-9fc4-acb66265226f - 748c0b5f a8ef 49a1 9fc4 acb66265226f

This is a summary of mean changes observed in a study comparing Metformin Hydrochloride Tablets to a placebo in pediatrics patients. The table shows the changes in Plasma Glucose and Body Weight at the final visit of the individuals. The Metformin Hydrochloride Tablets demonstrated a significant reduction in FPG levels with a p-value of less than 0.001 compared to the placebo. A slight increase in body weight was observed in both groups; however, the difference was not significant.*
c630784-d07a-495b-95e7-b3de307bd6c8 - 9c630784 d07a 495b 95e7 b3de307bd6c8

This is a table that summarizes the results of a 23-week study comparing the combined treatment of Metformin Hydrochloride Tablets and Glyburide to monotherapies of Glyburide or Metformin Hydrochloride Tablets in terms of the mean changes in fasting plasma glucose, HbA1c and body weight from baseline. The results show the p-values for Glyburide vs MET and MET vs Comb. All patients in the study were on Glyburide at baseline, and the changes in most categories were not statistically significant except for body weight reduction in the combined treatment group compared to the monotherapies.*
2e2c0e5-1dbe-449d-885e-e3a329b03f2e - d2e2c0e5 1dbe 449d 885e e3a329b03f2e

The table shows a comparison between the mean changes from baseline in HbA and daily insulin dose for a group treated with combined metformin hydrochloride tablets/insulin and another group treated with placebo insulin. The treatment difference is also highlighted. The study shows a statistically significant difference in the final visit for HbA using analysis of covariance with baseline as covariate (p=0.04) but not for insulin.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.