Product Images Losartan Potassium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Losartan Potassium NDC 68071-4823 by Nucare Pharmaceuticals,inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PDP - 68071 4823 6

This is a description of a medication called Losartan Potassium 50mg from NuCare Pharmaceuticals, Inc. The medication comes in a bottle with 60 tablets, and each film-coated tablet contains Losartan Potassium USP 50mg. The lot number for the medication is 000000, and the expiration date is 00-00. The tablet is oval-shaped, green, scored, and debossed with 'E' on one side, '4' and '6' on the scored side. The manufacturer's NDC is 57237-205-99. The medication should be stored at a controlled temperature of 59-86°F and kept out of reach of children. It is important to call your doctor for medical advice about any side effects, and you may also report side effects to the FDA.*
Figure 1 - losartan fig1

The text describes a chart of two treatments, Atenolol and Losartan Potassium, showing the percentage of patients with primary endpoint at different study months. It also indicates an adjusted risk reduction of 13% with a p-value of 0.021.*
Figure 2 - losartan fig2
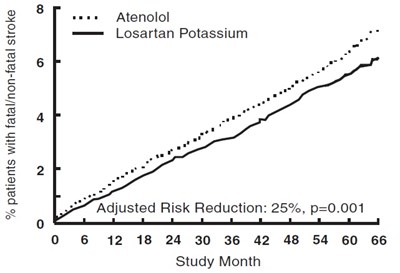
This is a study report that seems to involve the drugs Atenolol and Losartan Potassium. The report indicates a percentage of patients with either fatal/non-fatal results. Additionally, the study shows an adjusted risk reduction of 25% with a p-value of 0.001. The study monitored patients for 66 months.*
Figure 3 - losartan fig3
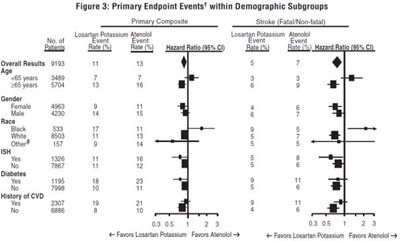
This is a table showing primary endpoint events within demographic subgroups, numbered by age, gender, race, and history of VD. Overall results are provided, but it is unclear what the primary endpoint events refer to.*
Figure-4 - losartan fig4

This appears to be a table or chart with some statistical analysis regarding the effectiveness of Losttan Potassium compared to a placebo. The table shows that 60% of patients with the condition experienced an event with Losttan Potassium, while 40% experienced an event with the placebo. There is a risk reduction percentage of 16.1% and a p-value of 0.022. However, without further context, it is difficult to provide a more detailed description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
