Product Images Atorvastatin Calcium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Atorvastatin Calcium NDC 68071-5188 by Nucare Pharmaceuticals,inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PDP - 68071 5188 9

The text is a product label for Atorvastatin Calcium 20mg tablets from NuCare Pharmaceuticals, Inc. It includes information such as the NDC number, lot number, expiration date, and serial number. It also provides a warning to keep the medication out of reach of children and instructions to store it at a controlled temperature of 68-77°F. There is a reference to the manufacturer's label for a full list of ingredients and a recommendation to call a doctor for medical advice about side effects.*
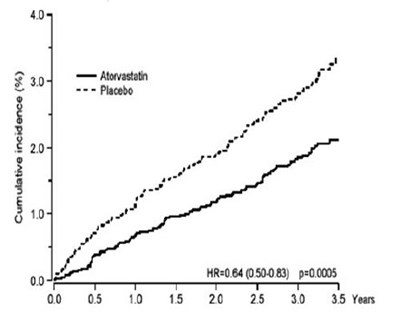
figure-02 - atorvastatin figure 02

This document provides information on the cumulative hazard expressed as percentage. It also includes an HR (hazard ratio) value of 0.63, with a confidence interval of 0.48-0.83, and a p-value of 0.001. The graph shows the time to reach the first primary endpoint over four (4) years of follow-up.*
figure-03 - atorvastatin figure 03
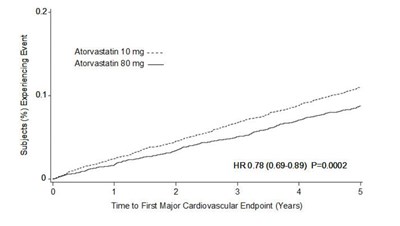
This text appears to be a table showing the percentage of subjects experiencing an event (presumably related to cardiovascular health) while taking Atorvastatin at two different doses. The 80 mg dose had a statistically significant decrease in the occurrence of the event compared to the 10 mg dose. The table also includes a graphical representation of the time to the first major cardiovascular endpoint over four years.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.