Product Images Enoxaparin Sodium
View Photos of Packaging, Labels & Appearance
- enoxaparin-spl-blister-label-100mg - enoxaparin spl blister label 100mg
- enoxaparin-spl-blister-label-120mg - enoxaparin spl blister label 120mg
- enoxaparin-spl-blister-label-150mg - enoxaparin spl blister label 150mg
- enoxaparin-spl-blister-label-30 mg - enoxaparin spl blister label 30 mg
- enoxaparin-spl-blister-label-40-mg - enoxaparin spl blister label 40 mg
- enoxaparin-spl-blister-label-60mg - enoxaparin spl blister label 60mg
- enoxaparin-spl-blister-label-80mg - enoxaparin spl blister label 80mg
- enoxaparin-spl-carton-100mg - enoxaparin spl carton 100mg
- enoxaparin-spl-carton-120mg - enoxaparin spl carton 120mg
- enoxaparin-spl-carton-150mg - enoxaparin spl carton 150mg
- enoxaparin-spl-carton-30 mg - enoxaparin spl carton 30 mg
- enoxaparin-spl-carton-40-mg - enoxaparin spl carton 40 mg
- enoxaparin-spl-carton-60mg - enoxaparin spl carton 60mg
- enoxaparin-spl-carton-80mg - enoxaparin spl carton 80mg
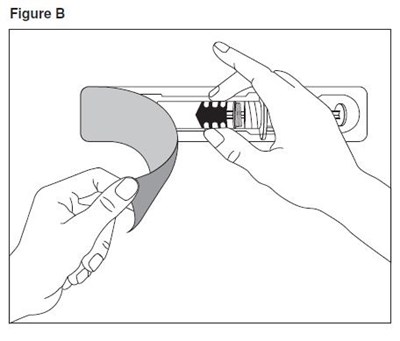
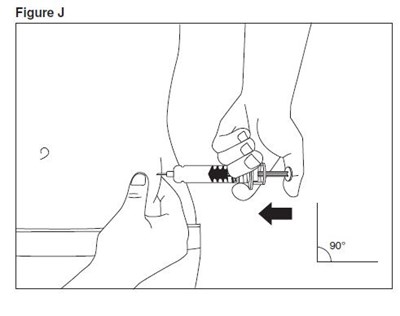
- enoxaparin-spl-figure1 - enoxaparin spl figure1
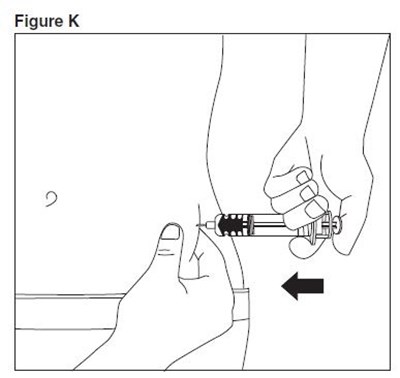
- enoxaparin-spl-figure10 - enoxaparin spl figure10
- enoxaparin-spl-figure11 - enoxaparin spl figure11
- enoxaparin-spl-figure12 - enoxaparin spl figure12
- enoxaparin-spl-figure13 - enoxaparin spl figure13
- enoxaparin-spl-figure14 - enoxaparin spl figure14
- enoxaparin-spl-figure15 - enoxaparin spl figure15
- enoxaparin-spl-figure16 - enoxaparin spl figure16
- enoxaparin-spl-figure17 - enoxaparin spl figure17
- enoxaparin-spl-figure18 - enoxaparin spl figure18
- enoxaparin-spl-figure19 - enoxaparin spl figure19
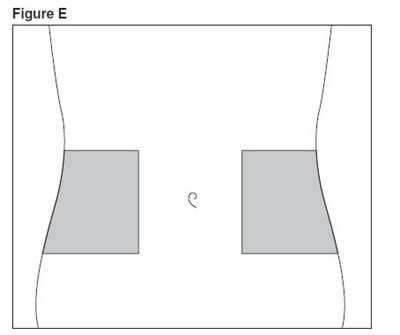
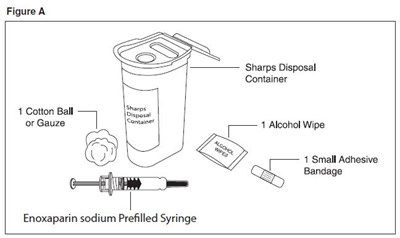
- enoxaparin-spl-figure2 - enoxaparin spl figure2
- enoxaparin-spl-figure20 - enoxaparin spl figure20
- enoxaparin-spl-figure21 - enoxaparin spl figure21
- enoxaparin-spl-figure22 - enoxaparin spl figure22
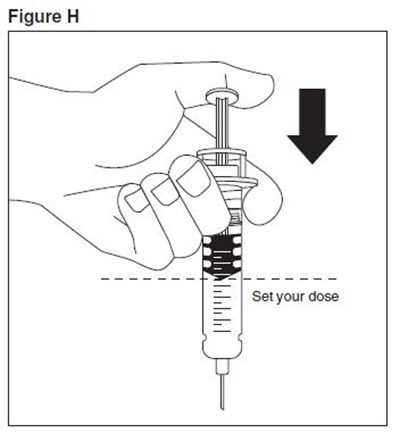
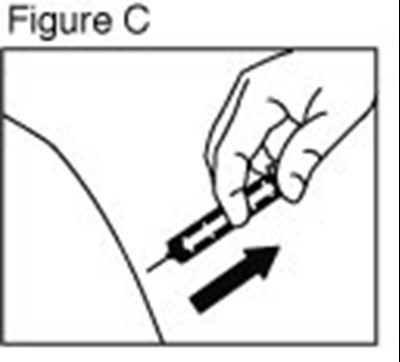
- enoxaparin-spl-figure3 - enoxaparin spl figure3
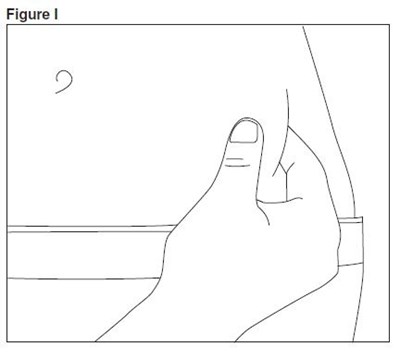
- enoxaparin-spl-figure4 - enoxaparin spl figure4
- enoxaparin-spl-figure5 - enoxaparin spl figure5
- enoxaparin-spl-figure6 - enoxaparin spl figure6
- enoxaparin-spl-figure7 - enoxaparin spl figure7
- enoxaparin-spl-figure8 - enoxaparin spl figure8
- enoxaparin-spl-figure9 - enoxaparin spl figure9
- enoxaparin-spl-pfs-label-100mg - enoxaparin spl pfs label 100mg
- enoxaparin-spl-pfs-label-120mg - enoxaparin spl pfs label 120mg
- enoxaparin-spl-pfs-label-150mg - enoxaparin spl pfs label 150mg
- enoxaparin-spl-pfs-label-30 mg - enoxaparin spl pfs label 30 mg
- enoxaparin-spl-pfs-label-40-mg - enoxaparin spl pfs label 40 mg
- enoxaparin-spl-pfs-label-60mg - enoxaparin spl pfs label 60mg
- enoxaparin-spl-pfs-label-80mg - enoxaparin spl pfs label 80mg
- enoxaparin-spl-structure - enoxaparin spl structure
- enoxaparin-spl-structure1 - enoxaparin spl structure1
Product Label Images
The following 45 images provide visual information about the product associated with Enoxaparin Sodium NDC 68083-638 by Gland Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
enoxaparin-spl-blister-label-150mg - enoxaparin spl blister label 150mg

This is a description of a medication called Enoxaparin Sodium Injection, USP. Each syringe contains 150 mg of the injection, derived from porcine intestinal mucosa. The medication is intended for subcutaneous (under the skin) injection. The dosage information and directions for use can be found in the package insert. The medication is manufactured by Gland Pharma Limited in Hyderabad, India. Each graduation on the syringe equals 3 mg of the injection. The syringe should be stored at a temperature of 20° to 25°C (68° to 77°F). It is a single dose syringe with an automatic safety device to prevent accidental needlesticks. Keep the medication out of reach of children. The syringe has a total volume of 1 mL.*
enoxaparin-spl-blister-label-30 mg - enoxaparin spl blister label 30 mg

This is a description for a medication called Enoxaparin Sodium. It is a single-dose syringe with an automatic safety device used for subcutaneous injection. Each syringe contains 30 mg of Enoxaparin Sodium derived from porcine intestinal mucosa. The medication is manufactured by Gland Pharma Limited and should be stored at a temperature of 20°C to 25°C. The package should be kept out of reach of children. For dosage information and directions for use, refer to the package insert.*
enoxaparin-spl-blister-label-40-mg - enoxaparin spl blister label 40 mg

This text appears to be a description of a medication called Enoxaparin Sodium Injection. It is indicated for subcutaneous injection and contains 40 mg of enoxaparin sodium derived from porcine intestinal mucosa. The medication is manufactured by Gland Pharma Limited in Hyderabad, India. The package insert should be consulted for dosage information and directions for use. The syringe comes with an automatic safety device. The medication should be stored at controlled room temperature between 20° to 25°C (68° to 77°F). It is important to keep this medication out of reach of children.*
enoxaparin-spl-blister-label-80mg - enoxaparin spl blister label 80mg

This is a description of a medication called Enoxaparin Sodium Injection, USP. It comes in a single dose syringe with an automatic safety device for subcutaneous injection. Each syringe contains 80 mg of Enoxaparin Sodium derived from porcine intestinal mucosa in Water for Injection. The dosage information and directions for use can be found in the package insert. The medication is manufactured by Gland Pharma Limited in India. Each graduation on the syringe equals 2.5 mg. It is recommended to store the medication at 20° to 25°C (68° to 77°F). There is a warning to keep it out of reach of children.*
enoxaparin-spl-pfs-label-120mg - enoxaparin spl pfs label 120mg

This is a description of a medication called Enoxaparin Sodium Injection, USP. It is a 120 mg/0.8 mL solution intended for subcutaneous injection. It is manufactured by Gland Pharma Limited. The lot and expiry dates are not available in the provided text. The product is packaged in a 17 x 6 mm size container. The text also includes a product code: NDC 68083-639-01.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.