Product Images Voxzogo 0.4mg
View Photos of Packaging, Labels & Appearance
- Chemical Structure - voxzogo 01
- Image - voxzogo 02
- Image - voxzogo 03
- Image - voxzogo 04
- Image - voxzogo 05
- Image - voxzogo 06
- Image - voxzogo 07
- Image - voxzogo 08
- Step 1 - voxzogo 09
- Step 2 - voxzogo 10
- Step 3 - voxzogo 11
- Step 4 - voxzogo 12
- Step 5 - voxzogo 13
- Step 6 - voxzogo 14
- Step 7 - voxzogo 15
- Step 8 - voxzogo 16
- Step 9 - voxzogo 17
- Step 10 - voxzogo 18
- Step 11 - voxzogo 19
- Step 12 - voxzogo 20
- Step 13 - voxzogo 21
- Step 14 - voxzogo 22
- Step 15 - voxzogo 23
- Step 16 - voxzogo 24
- Step 17 - voxzogo 25
- Step 17 - voxzogo 26
- Image - voxzogo 27
- Step 18 - voxzogo 28
- PRINCIPAL DISPLAY PANEL - Kit Carton - 68135-082 - voxzogo 29
- PRINCIPAL DISPLAY PANEL - 0.4 mg Vial Label - voxzogo 30
- PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Label - voxzogo 31
- PRINCIPAL DISPLAY PANEL - Kit Carton - 68135-119 - voxzogo 32
- PRINCIPAL DISPLAY PANEL - 0.56 mg Vial Label - voxzogo 33
- PRINCIPAL DISPLAY PANEL - 0.7 mL Syringe Label - voxzogo 34
- PRINCIPAL DISPLAY PANEL - Kit Carton - 68135-181 - voxzogo 35
- PRINCIPAL DISPLAY PANEL - 1.2 mg Vial Label - voxzogo 36
- PRINCIPAL DISPLAY PANEL - 0.6 mL Syringe Label - voxzogo 37
Product Label Images
The following 37 images provide visual information about the product associated with Voxzogo 0.4mg NDC 68135-082 by Biomarin Pharmaceutical Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image - voxzogo 02

This text lacks context to provide a description. It seems to be related to some sort of medication in the form of a powder, and there is mention of a cap, vial stopper, and vial, but without additional information, it is not possible to provide a useful description.*
Image - voxzogo 04

This appears to be a description of a needle labeled as "Diluent Needle" with a blue tab needle cap that has a retractable needle. The text also mentions that the needle is "safe" but it's not clear what that means without more context.*
Image - voxzogo 05

A container designed for the safe disposal of sharp objects and needles, typically used in medical or laboratory settings.*
Image - voxzogo 06

This is a prefilled syringe containing diluent with a plunger rod cap. It is meant to be used with BMN-111 and sterile water is used to reconstitute it.*
Image - voxzogo 08

This appears to be a basic description of an injection syringe, including components such as the needle, needle cap, plunger rod, and a reference to the needle being inside.*
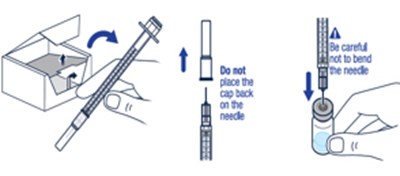
Step 1 - voxzogo 09

This text is a warning about avoiding touching the stopper of a vial after wiping it with an alcohol pad.*
Step 5 - voxzogo 13

This text seems to be a cautionary message instructing users to avoid touching the needle tip and to press the blue tab to retract the needle. The character '¢' may have been a symbol representing a certain action but could not be interpreted due to errors.*
Step 8 - voxzogo 16

The text provides some brief instructions about handling a vial, warning the reader to be cautious not to bend the needle while doing so. It is not clear what specific context or situation these instructions refer to.*
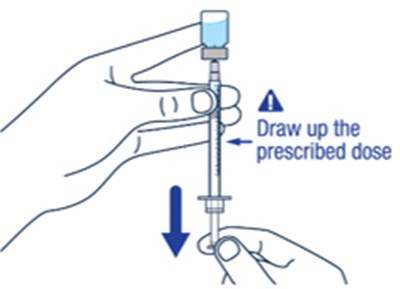
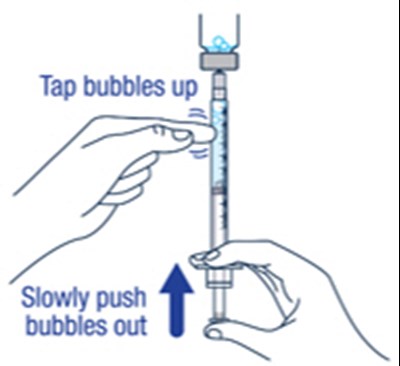
Step 11 - voxzogo 19

This text provides instructions on two important aspects of administering medication, namely, removing bubbles from the syringe and measuring the prescribed dose accurately. It advises the reader to remove any large bubbles from the syringe and states that some small bubbles are acceptable. Additionally, it emphasizes the importance of ensuring that the dose in the syringe matches the prescribed dose.*
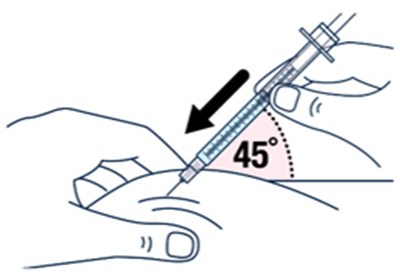
Step 13 - voxzogo 21

This text gives instructions about proper injection sites for Voxzogo. It advises against injecting through clothes or into skin that is swollen, sore, bruised, or scared. The recommended injection sites are thighs (not through clothes), abdomen (2 inches from belly button), buttocks, and the back of the upper arms. This text may be useful for healthcare providers or caregivers administering Voxzogo injections.*
PRINCIPAL DISPLAY PANEL - 0.4 mg Vial Label - voxzogo 30

This is a pharmaceutical label for a single-dose vial with the drug code MLC7246800. The label advises to reconstitute prior to use and that it is for subcutaneous use only. The drug is available only by prescription. The label also includes a LOT and EXP number for tracking the drug's expiration date.*
PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Label - voxzogo 31

This is a description for the drug diluent used for VOXZOGOTM injection. It should only be used for dilution as directed and is single-dose with no preservatives. The product is distributed by BioMarin and includes a lot number and expiration date.*
PRINCIPAL DISPLAY PANEL - 0.7 mL Syringe Label - voxzogo 34

This is a drug diluent for VOXZOGOT™ for Injection, with NDC code 68135-102-43 and is distributed by BioMarin. It is a single-dose diluent with no preservative and must be reconstituted as directed. The lot number is 10008843-02 with an expiration date (EXP) that is not available in the given text.*
PRINCIPAL DISPLAY PANEL - 1.2 mg Vial Label - voxzogo 36

This is a medication called VOXZOoGOo™ or vosoritide, which is available in a single-dose vial for subcutaneous injection only. The vial contains 1.2 mg of the medication, and it needs to be reconstituted before use. It is manufactured by a company called BioMarin.*
PRINCIPAL DISPLAY PANEL - 0.6 mL Syringe Label - voxzogo 37

This is a description of a diluent for VOXZOGOTM injection. It is available by prescription only and should only be used as a drug diluent. The diluent is single-dose and does not contain any preservatives. It is distributed by BioMarin and comes with a lot and expiration number.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.