FDA Label for Nitro-time
View Indications, Usage & Precautions
Nitro-time Product Label
The following document was submitted to the FDA by the labeler of this product Carilion Materials Management. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Description:
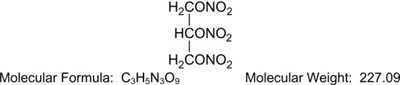
Nitroglycerin is 1,2,3-propanetriol trinitrate, an organic nitrate whose structural formula is:
The organic nitrates are vasodilators, active on both arteries and veins. Each Extended-Release Capsule, for oral administration contains 2.5 mg, 6.5 mg, or 9 mg of Nitroglycerin.
The inactive ingredients in each capsule are corn starch, ethylcellulose, gelatin, lactose monohydrate, pharmaceutical glaze, sugar, talc, and wax. Additionally the 2.5 mg capsule contains FD&C Blue #1, D&C Yellow #10, FD&C Red #40, D&C Red #28; the 6.5 mg capsule contains D&C Yellow #10, FD&C Yellow #6, FD&C Blue #1, D&C Red #33; the 9 mg capsule contains D&C Yellow #10, FD&C Yellow #6, FD&C Green #3, and titanium dioxide.
Clinical Pharmacology
The principal pharmacological action of nitroglycerin is relaxation of vascular smooth muscle and consequent dilation of peripheral arteries and veins, especially the latter. Dilatation of the veins promotes peripheral pooling of blood and decreases venous return to the heart, thereby reducing left ventricular end-diastolic pressure and pulmonary capillary wedge pressure (preload). Arteriolar relaxation reduces systemic vascular resistance, systolic arterial pressure, and mean arterial pressure (afterload). Dilatation of the coronary arteries also occurs. The relative importance of preload reduction, afterload reduction, and coronary dilatation remains undefined. Dosing regimens for most chronically used drugs are designed to provide plasma concentrations that are continuously greater than a minimally effective concentration. This strategy is inappropriate for organic nitrates. Several well-controlled clinical trials have used exercise testing to assess the anti-anginal efficacy of continuously-delivered nitrates. In the large majority of these trials, active agents were indistinguishable from placebo after 24 hours (or less) of continuous therapy. Attempts to overcome nitrate tolerance by dose escalation, even to doses far in excess of those used acutely, have consistently failed. Only after nitrates had been absent from the body for several hours was their anti-anginal efficacy restored.
Indications And Usage
Nitroglycerin Extended-Release Capsules are indicated for the prevention of angina pectoris due to coronary artery disease. The onset of action of oral nitroglycerin is not sufficiently rapid for this product to be useful in aborting an acute anginal episode.
Contraindications
Allergic reactions to organic nitrates are extremely rare, but they do occur. Nitroglycerin is contraindicated in patients who are allergic to it.
Warnings
The benefits of oral nitroglycerin in patients with acute myocardial infarction or congestive heart failure have not been established. If one elects to use nitroglycerin in these conditions, careful clinical or hemodynamic monitoring must be used to avoid the hazards of hypotension and tachycardia.
Because the effects of capsules are so difficult to terminate rapidly, they are not recommended in these settings.
Adverse Reactions
Adverse reactions to nitroglycerin are generally dose-related, and almost all of these reactions are the result of nitroglycerin's activity as a vasodilator. Headache, which may be severe, is the most commonly reported side effect. Headache may be recurrent with each, daily dose, especially at higher doses. Transient episodes of lightheadedness, occasionally related to blood pressure changes, may also occur. Hypotension occurs infrequently, but in some patients it may be severe enough to warrant discontinuation of therapy. Syncope, crescendo angina, and rebound hypertension have been reported but are uncommon.
Allergic reactions to nitroglycerin are also uncommon, and the great majority of those reported have been cases of contact dermatitis or fixed drug eruptions in patients receiving nitroglycerin in ointments or patches. There have been a few reports of genuine anaphylactoid reactions, and these reactions can probably occur in patients receiving nitroglycerin by any route.
Extremely rarely, ordinary doses of organic nitrates have caused methemoglobinemia in normal-seeming patients; for further discussion of its diagnosis and treatment, see . OVERDOSAGE
Data are not available to allow estimation of the frequency of adverse reactions during treatment with Nitroglycerin Extended-Release Capsules.
Dosage And Administration:
As noted above ( ) careful studies with other formulations of nitroglycerin have shown that maintenance of continuous 24-hour plasma levels of nitroglycerin results in tolerance (i.e., loss of clinical response). Every dosing regimen for Nitroglycerin Extended-Release Capsules should provide a daily nitrate-free interval to avoid the development of this tolerance. The minimum necessary length of such an interval has not been defined, but studies with other nitroglycerin formulations have shown that 10 to 12 hours is sufficient. Large controlled studies with other formulations of nitroglycerin show that no dosing regimen with Nitroglycerin Extended-Release Capsules should be expected to provide more than about 12 hours of continuous anti-anginal efficacy per day. CLINICAL PHARMACOLOGY
The pharmacokinetics of Nitroglycerin capsules, and the clinical effects of multiple-dose regimens, have not been well studied. In clinical trials, the initial regimen of Nitroglycerin has been 2.5 to 6.5 mg three to four times a day, with subsequent upward dose adjustment guided by symptoms and side effects. In one trial, 5 of the 18 subjects were titrated up to a dose of 26 mg four times a day.
How Supplied
NDC:68151-2503-6 in a PACKAGE of 1 CAPSULES
Storage
Store at controlled room temperature 15° - 30° C (59° - 86° F).
Dispense in a tight container, as defined in the USP.
Manufactured by: Distributed by:
Time Cap Labs, Inc. Major Pharmaceuticals 7 Michael Avenue 31778 Enterprise Drive Farmingdale, NY 11735 Livonia, MI 48150 USA
Revised March 2005
* Please review the disclaimer below.