Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
- str - 94fb4253 7822 4043 8ef9 0f8d93642feb 01
- fig.1 - 94fb4253 7822 4043 8ef9 0f8d93642feb 02
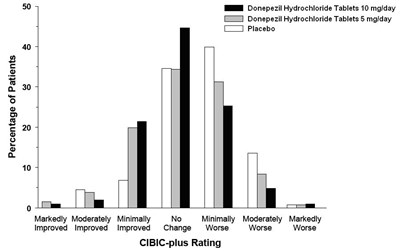
- Figure 2 - 94fb4253 7822 4043 8ef9 0f8d93642feb 03
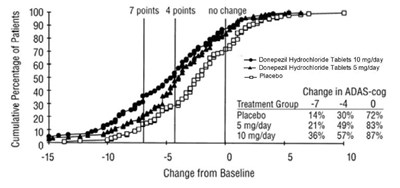
- Figure 3 - 94fb4253 7822 4043 8ef9 0f8d93642feb 04
- 5 - 94fb4253 7822 4043 8ef9 0f8d93642feb 05
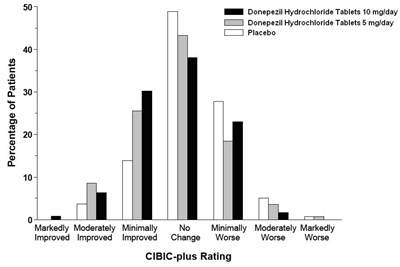
- figure 5 - 94fb4253 7822 4043 8ef9 0f8d93642feb 06
- figure 6 - 94fb4253 7822 4043 8ef9 0f8d93642feb 07
- Figure 7 - 94fb4253 7822 4043 8ef9 0f8d93642feb 08
- Figure 8 - 94fb4253 7822 4043 8ef9 0f8d93642feb 09
- Figure 9 - 94fb4253 7822 4043 8ef9 0f8d93642feb 10
- Figure 10 - 94fb4253 7822 4043 8ef9 0f8d93642feb 11
- logo - 94fb4253 7822 4043 8ef9 0f8d93642feb 12
- logo - 94fb4253 7822 4043 8ef9 0f8d93642feb 13
- Label Image - lbl681513114
Product Label Images
The following 14 images provide visual information about the product associated with Donepezil Hydrochloride NDC 68151-3114 by Carilion Materials Management, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
fig.1 - 94fb4253 7822 4043 8ef9 0f8d93642feb 02
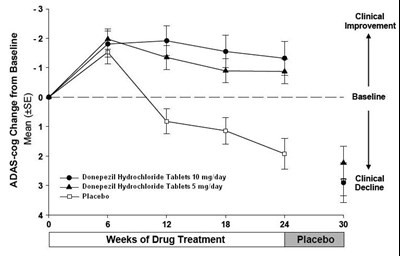
The text provides a table showing the mean change in ADAS-cog score from baseline to 6, 12, and 18 weeks of drug treatment with Donepezil Hydrochloride Tablets at 5mg and 10mg, versus placebo. There is also reference to "Clinical Improvement" and "Clinical Decline". No further information is available.*
Figure 2 - 94fb4253 7822 4043 8ef9 0f8d93642feb 03
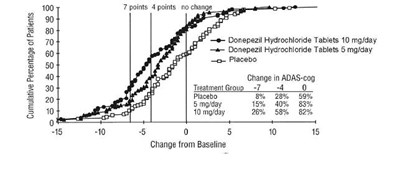
This is a graph displaying the cumulative percentage of patients receiving Donepezil Hydrochloride Tablets at different dosages and also the placebo, based on their change in ADAS-cog score. The ADAS-cog score is a measure of cognitive function in Alzheimer's disease patients. The graph shows that patients receiving the higher dose of Donepezil Hydrochloride Tablets (10 mg/day) experienced greater improvement in cognitive function compared to those receiving a lower dose (5 mg/day) or placebo. The x-axis shows the change from baseline (initial ADAS-cog score) and the y-axis shows the cumulative percentage of patients.*
5 - 94fb4253 7822 4043 8ef9 0f8d93642feb 05
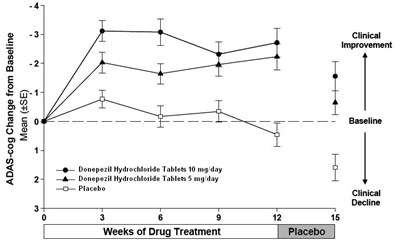
This is a graph that shows the change from baseline in ADAS-cog (a test for assessing cognitive function in patients with Alzheimer's disease) over 12 weeks of drug treatment with either Donepezil Hydrochloride Tablets or a Placebo. The y-axis represents the mean (+SE) change in ADAS-cog score, while the x-axis represents the treatment period in weeks. The graph shows that both dosages of Donepezil Hydrochloride Tablets resulted in clinical improvement over the 12 weeks, while the Placebo group experienced clinical decline in cognitive function.*
Figure 8 - 94fb4253 7822 4043 8ef9 0f8d93642feb 09
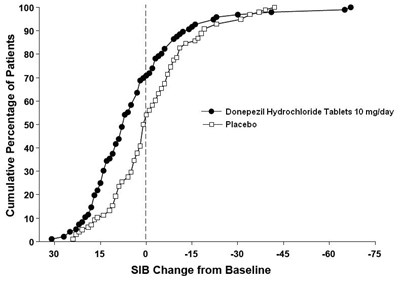
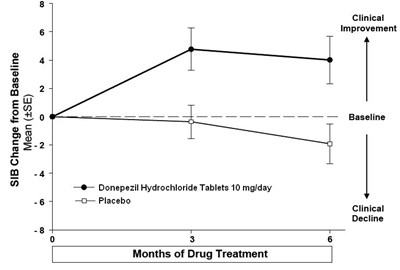
The text provides a graph showing the cumulative percentage of patients and the SIB (Severe Impairment Battery) change from baseline for Donepezil Hydrochloride Tablets 10mg/day and Placebo. The graph shows that Donepezil Hydrochloride Tablets have a positive effect compared to a Placebo in improving SIB scores.*
Figure 9 - 94fb4253 7822 4043 8ef9 0f8d93642feb 10
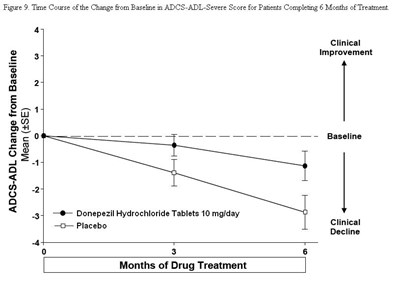
This is a graph that shows the time course of change from baseline in ADCS-ADL-Severe score for patients who completed 6 months of treatment. The x-axis represents the time in months, while the y-axis represents the ADCS-ADL-Severe score. There are two lines: one for patients who received donepezil hydrochloride tablets 10 mg/day and one for patients who received a placebo. The graph suggests that patients who received donepezil hydrochloride tablets experienced some clinical improvement. However, a detailed description of the results cannot be given due to lack of context.*
Figure 10 - 94fb4253 7822 4043 8ef9 0f8d93642feb 11

This appears to be a graph with the title "Cumulative Percentage of Patients" with a Y-axis starting at 0 and ending at 100. The X-axis has a scale from 0-100 with marks at 15, 10, 5, 0, -5, and -10. The graph itself has two lines - one labeled "Donepezil Hydrochloride Tablets 10 mg/day" and the other labeled "Placebo" - both running from around 20% at the left side of the graph to 100% at the right side. Below the graph is a label reading "ADCS-ADL Change from Baseline".*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.