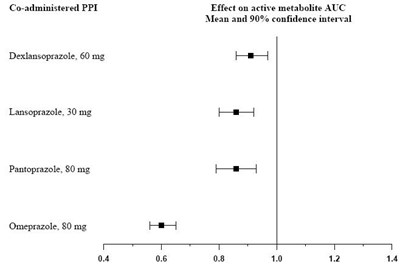
Product Images Clopidogrel Bisulfate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Clopidogrel Bisulfate NDC 68151-3118 by Carilion Materials Management, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure2 - figure2

This is a chart comparing the cumulative event rate for patients taking placebo with aspirin versus clopidogrel with aspirin after several months of follow-up. Other standard therapies were also used as needed. The purpose and nature of the events recorded are not available.*
figure3 - figure3

This appears to be a table depicting different types of events and their percentage in different treatments or groups, possibly related to medical treatments. The table includes information such as the total number of events, the percentage of events, and the specific group or treatment associated with them. There also seems to be a disclaimer or note at the end stating that other standard therapies were used as appropriate.*
figure4 - figure4

This appears to be a medical study report that compares the effects of placebo and clopidogrel on reducing the risk of death. According to the results shared, clopidogrel was associated with a 7% proportional risk reduction in comparison to placebo. The study followed patients for up to 28 days and the number of deaths reported for each group is shared.*
figure5 - figure5

The given text appears to be data from a study on the effectiveness of a drug called Clopidogrel in reducing the risk of death, reinfarction (recurrence of heart attack), or stroke in patients before their first discharge. The study involved two groups - one that was administered Clopidogrel and the other given a placebo. The data shows that out of the 2310 patients who were given a placebo, 10.1% experienced an event (death, reinfarction, or stroke) whereas out of the 2121 patients who were administered Clopidogrel, only 9.2% experienced an event. This suggests that there was a 9% proportional risk reduction in the group that received Clopidogrel. The study also measured the number of days since randomization up to 28 days.*
Label Image - lbl681513118

This is a medication called Clopidogrel, which comes in tablet form with a dosage strength of 75 milligrams. The manufacturer of the drug is Sun Pharma and the lot number is JKM6120A. The product's expiration date is August 2014, and there is a code printed on the tablet as W0225143m2.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.