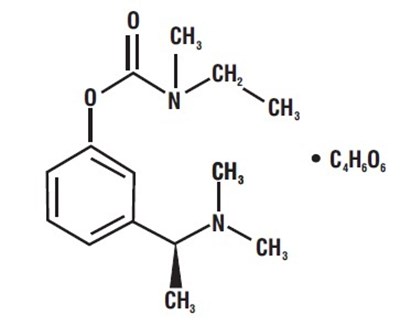
Product Images Rivastigmine Tartrate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Rivastigmine Tartrate NDC 68151-4134 by Carilion Materials Management, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure1 - figure1

The text contains numerical and alphabetic data, which seems to be related to a scientific study or clinical trial. The figure presented shows the time-course change from the baseline in ADAS-cog score for patients completing 26 weeks of treatment in Study 1. However, there is not enough context to provide a detailed description or analysis.*
figure2 - figure2

This is a graph showing the percentage of patients who completed 26 weeks of double-blind treatment with specified changes from baseline ADAS-cog scores. The X-axis represents the change from the baseline ADAS-cog score, and the Y-axis represents the cumulative percentage of patients. The graph shows three treatment groups: placebo, 1 mg to 4 mg, and 6 mg to 12 mg, and the percentages of randomized patients who completed the study. The placebo group had an 84% completion rate, the 1 mg to 4 mg group had an 85% completion rate, and the 6 mg to 12 mg group had a 65% completion rate.*
figure3 - figure3

The figure depicts the frequency distribution of CIBIC-Plus scores at Week 26 in Study 1. The data is represented through a graph that shows the percentage of patients grouped by improvement level of their condition. The improvement levels are described as Markedly Improved, Moderately Improved, Minimally Improved, Unchanged, Minimally Worse, Moderately Worse, and Markedly Worse. The graph includes two treatment groups, placebo and doses of 1 to 4mg and 6 to 12mg.*
figure4 - figure4

This is a graph showing the change from the baseline in ADAS-cog score over 26 weeks of treatment. The graph indicates clinical improvement and decline for patients completing the treatment. There are different dosages indicated at the top of the graph. There is no further information available.*
figure5 - figure5

The text provides information about the cumulative percentage of patients completing 26 weeks of double-blind treatment with specified changes from baseline ADAS-cog scores. There are different treatment groups with varying completion rates. The figure shows a graph with the change in ADAS-cog scores for each treatment group. The text does not provide details about the type of condition being treated or the methodology used, making it difficult to interpret the findings.*
figure7 - figure7

The figure shows the time course of the change from baseline in ADAS-cog score for patients who completed 24 weeks of treatment in Study 4. The mean change from baseline is presented using standard error of the mean (SEM) for rivastigmine tartrate and placebo groups. The ADAS-cog rating scale is used to assess cognitive function in Alzheimer's disease. This information is useful for healthcare professionals and researchers studying the efficacy of rivastigmine tartrate in treating Alzheimer's disease.*
Label Image - lbl681514134

This is a medication called Rivastigmine Tartrate, in the form of a 1.5mg capsule, manufactured by SUN. The lot number is JKM1949A and expiration date is 07/14, which suggests the medication may be outdated or expired. The text also includes a prescription code "Rx only" and a number "24134". However, the last line appears to be garbled and not readable.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.