Product Images Levetiracetam
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Levetiracetam NDC 68180-113 by Lupin Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 01

This text provides a formula for calculating the total daily dose of a medication, based on the patient's weight and the daily dose prescribed in milligrams per kilogram. The medication appears to be in a liquid form with a concentration of 100 milligrams per milliliter.*
e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 02

This text contains a formula for estimating renal clearance (CLer) in patients. The formula varies for male and female patients based on their weight and age. For female patients, CLer is computed using (140-age) x weight (kg) divided by 72 x serum creatinine (mg/dL). After which, the result is adjusted to account for the patient's Body Surface Area (BSA). For males, the same formula is used but with an additional constant factor of 1.23.*
e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 05

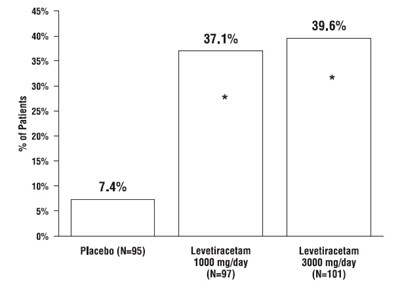
This appears to be a table showing the percentages of patients in different groups receiving different dosages of levetiracetam or a placebo. However, without further context, it is difficult to determine the purpose or results of this table.*
e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 08

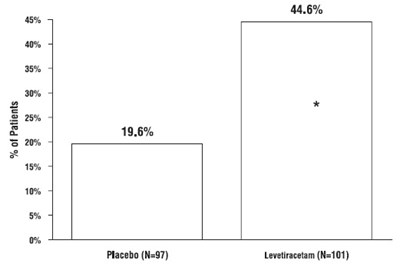
This is a summary of a clinical trial or study that included a placebo group (51 patients) and a group treated with levetiracetam (58 patients). The percentage of patients who experienced a certain outcome is provided for each group, but the nature of the outcome is not specified.*
e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 09

This appears to be a chart showing the percentage of patients experiencing a certain outcome while taking either a placebo or Levetiracetam. The chart shows the percentage of patients on a vertical scale ranging from 0% to 100%, with intervals of 10%. The horizontal axis shows the two treatment groups, Placebo (N=84) and Levetiracetam (N=79), with the percentages of patients in each group experiencing the outcome represented as bars. However, there is not enough information to determine what the outcome being measured is.*
e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 10

Levetiracetam Tablets USP with NDC number 68180-112-02, is a prescription medication dispensed by pharmacists in a tightly sealed and light-resistant container with child-resistant closure. Each film-coated tablet contains levetiracetam USP 250 mg with usual dosage as outlined in the package insert for complete dosage recommendations. pharmacy dispenses it to patients with a medication guide. The medication should be stored at an average temperature of 25°C (77°F) and can be kept at excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Lupin Pharmaceuticals, Inc in Baltimore, Maryland 21202 is the company that produces LUPIN 500 Tablets. Med guide is available on www.lupin.com/levetiracetamtab- or can be seen by scanning the QR code.*
e24dd8a7 86c4 4979 8f2e da2d82f5d6d8 13

This is a description of a medication called Levetiracetam packaged by pharmaceutical company Lupin. The tablets come in 1000mg strength and are dispensed with a Medication Guide to each patient. The Pharmacist should store it in a tight light-resistant container with child-resistant closure. Complete dosage recommendations can be found in the package insert. The medication should be stored at 25°C (77°F) and excursions up to 15-30°C. The Medication Guide can be found on the Lupin website or by scanning the QR code. The manufacturer is Lupin Limited in Nagpur, India.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.