Product Images Perindopril Erbumine
View Photos of Packaging, Labels & Appearance
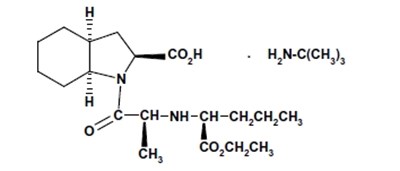
- Perindopril erbumine structural formula - fbb31482 9ac8 48c6 b814 5d467f22051b 01
- Figure 1. - fbb31482 9ac8 48c6 b814 5d467f22051b 02
- Figure 2 - fbb31482 9ac8 48c6 b814 5d467f22051b 03
- Perindopril Erbumine Tablets, 2 mg. - fbb31482 9ac8 48c6 b814 5d467f22051b 04
- Perindopril Erbumine Tablets, 4 mg. - fbb31482 9ac8 48c6 b814 5d467f22051b 05
- Perindopril Erbumine Tablets, 8 mg. - fbb31482 9ac8 48c6 b814 5d467f22051b 06
Product Label Images
The following 6 images provide visual information about the product associated with Perindopril Erbumine NDC 68180-237 by Lupin Pharmaceuticals, Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. - fbb31482 9ac8 48c6 b814 5d467f22051b 02

The text describes a graph showing the results of a clinical trial comparing the effectiveness of Placebo and Perindopril treatment over a period of four years. The y-axis shows a logarithmic scale for p-values ranging from 0 to 0.001. The x-axis shows the time in years, labeled from 0 to 4. The plot shows two distinct curves representing the Placebo and Perindopril treatments. The plot suggests that the Perindopril treatment is significantly more effective than the Placebo treatment, with a p-value of 0.0003. The number of patients at risk for both treatments is provided for each year.*
Figure 2 - fbb31482 9ac8 48c6 b814 5d467f22051b 03

This appears to be a tabular representation of the distribution of primary events (%), along with the number of patients, their gender, age, and various medical conditions such as hypertension, diabetes, and the use of certain drugs. The table also compares the effectiveness of perindopril versus a placebo in terms of "favors" or positive outcomes for each of the medical conditions.*
Perindopril Erbumine Tablets, 2 mg. - fbb31482 9ac8 48c6 b814 5d467f22051b 04

This is a description of a medication called Perindopri, which comes in tablet form and has a NDC number of 68180-255-0. The dosage and administration details can be found on the accompanying prescribing information. It is important to keep these tablets out of the reach of children, and store them at a temperature between 20°C to 25°C, while protecting them from moisture. The tablets are manufactured by Lupin Limited in Goa, India, and are distributed in the United States by Lupin Pharmaceuticals, Inc. Each tablet contains 2mg of perindopril erbumine.*
Perindopril Erbumine Tablets, 4 mg. - fbb31482 9ac8 48c6 b814 5d467f22051b 05

This is a drug information sheet for Perindopril Erbumine Tablets, containing the drug's National Drug Code (NDC: 68180-256-01), dosage and administration instructions, and manufacturer's information. The tablets contain 4 mg of perindopril erbumine and should be stored at 20-25°C to protect from moisture. The drug is manufactured by Lupin Pharmaceuticals, Inc. and is only available with a prescription.*
Perindopril Erbumine Tablets, 8 mg. - fbb31482 9ac8 48c6 b814 5d467f22051b 06

This is a prescription drug with NDC number 68180-237-01, which contains 8 mg of perindopril erbumine per tablet. It is manufactured by Lupin Limited in Goa, India and distributed by Lupin Pharmaceuticals, Inc. in Baltimore, Maryland. Dosage and administration information is available in the prescribing information. It should be stored at controlled room temperature and protected from moisture, and kept out of reach of children.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.