Product Images Divalproex Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Divalproex Sodium NDC 68180-260 by Lupin Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image 2 - d95f9b1b f1e4 4ae4 a215 7317fb1ca565 02

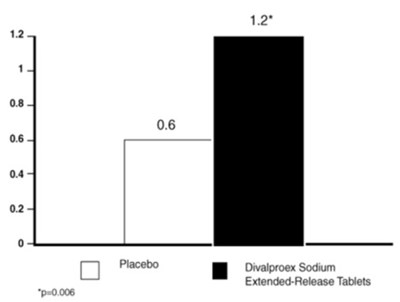
This text appears to be a graph or chart showing the reduction in CPS (presumably referring to some sort of medical measurement) rate when patients are treated with Divalproex Sodium Delayed-Release compared to a placebo. The graph shows that there is a 50% improvement in patients. However, without more information and context, it is difficult to provide a more detailed description.*
Image 5 - d95f9b1b f1e4 4ae4 a215 7317fb1ca565 05

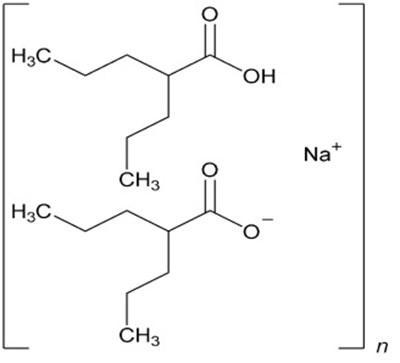
Each extended-release tablet of Divalproex Sodium contains valproic acid and is used to treat seizures and bipolar disorder. The usual dosage information is available in the accompanying prescribing information. The tablets must be stored in a tightly closed container at a controlled room temperature. Dispensing must be done with a medication guide provided separately to each patient. This drug is manufactured by Lupin in India and the code number is MH/DRUGS/25-ND/59.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.