Product Images Diclofenac Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
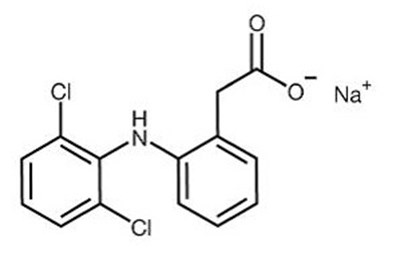
The following 8 images provide visual information about the product associated with Diclofenac Sodium NDC 68180-537 by Lupin Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image 1 - b385f9bc 6ae3 45a6 8e32 eb902dd89ed9 07

This is a description for a topical solution called Diclofenac Sodium, indicated for external use only. The usual dosage for affected knees is to apply two pump activations twice a day. Each pump actuation delivers 1 gram of solution containing 20 mg of ciclofenac sodium. It comes in a 3.8 FL.OZ. bottle and should be dispensed with an enclosed medication guide. After application, hands should be washed thoroughly to avoid contact with the eyes or mucous membranes. If persistent skin irritation occurs, the product should be discontinued and a physician should be consulted. The solution contains excipients such as dimethyl sulfoxide, dehydrated alcohol, hydroxypropylcellulose, propylene glycol, and purified water. It should be stored at controlled room temperature between 15°C and 30°C. The product is manufactured by Lupin Limited and distributed by Lupin Pharmaceuticals Inc. in Baltimore, Maryland, USA. The size of the product is 35 x 17 mm.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.