Product Images Lamivudine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Lamivudine NDC 68180-602 by Lupin Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image-02 - 86a45a3b a934 417d bc27 6d18fbff5f2d 02
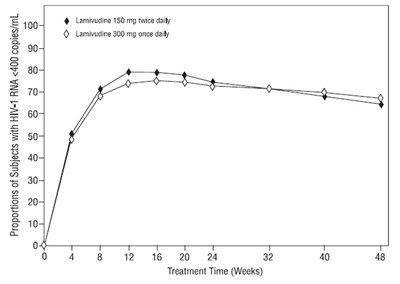
The text appears to be a graph showing the proportion of subjects with HIV-1 RNA less than 400 copies/mL over a period of 32 weeks. The graph compares two treatment options - Lamivudine 150mg twice daily and Lamivudine 300mg once daily.*
image-03 - 86a45a3b a934 417d bc27 6d18fbff5f2d 03

This is a prescription drug manufactured by Lupin Pharmaceuticals, Inc. NDC 68180-602-07 is the identification code for this particular drug. The drug contains 150 mg of lamivudine USP and comes in a container with 60 tablets. The recommended storage temperature is between 15°C to 30°C (59°F to 86°F). More information on the dosage can be found on the prescribing information accompanying the drug. The manufacturer is Lupin Limited and the drug code is GO/DRUGS/654.*
image-04 - 86a45a3b a934 417d bc27 6d18fbff5f2d 04

This is a medication called Lamivudine manufactured by Lupin Pharmaceuticals, Inc. It contains 300 mg of lamivudine per film-coated tablet, and should be stored at 25°C (77°F) with a permitted range of 15°C to 30°C (59°F to 86°F). The dosage information can be found in the accompanying prescribing information. The tablets are controlled by USP and Lupin Limited in Goa, India.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
