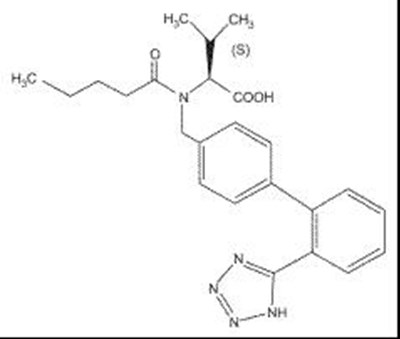
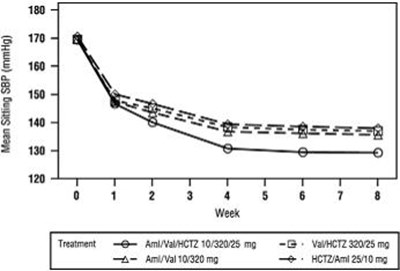
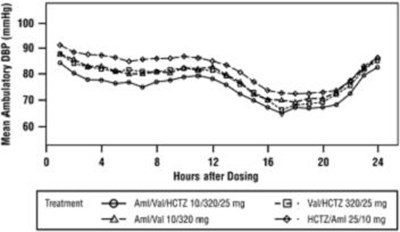
Product Images Amlodipine, Valsartan And Hydrochlorothiazide
View Photos of Packaging, Labels & Appearance
- 111922d9 0549 464b 8b1f d8240a5f7900 01
- 111922d9 0549 464b 8b1f d8240a5f7900 02
- 111922d9 0549 464b 8b1f d8240a5f7900 03
- 111922d9 0549 464b 8b1f d8240a5f7900 04
- 111922d9 0549 464b 8b1f d8240a5f7900 05
- 111922d9 0549 464b 8b1f d8240a5f7900 06
- 111922d9 0549 464b 8b1f d8240a5f7900 07
- 111922d9 0549 464b 8b1f d8240a5f7900 08
- 111922d9 0549 464b 8b1f d8240a5f7900 09
- 111922d9 0549 464b 8b1f d8240a5f7900 10
- 111922d9 0549 464b 8b1f d8240a5f7900 11
- 111922d9 0549 464b 8b1f d8240a5f7900 12
- 111922d9 0549 464b 8b1f d8240a5f7900 13
Product Label Images
The following 13 images provide visual information about the product associated with Amlodipine, Valsartan And Hydrochlorothiazide NDC 68180-774 by Lupin Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
111922d9 0549 464b 8b1f d8240a5f7900 09
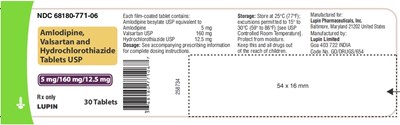
This is a description of a medication, but the only information provided is the NDC (National Drug Code) number, the dosage of Amlodipine and Hydrochlorothiazide, and storage instructions. There is no additional information about the medication, its uses or side effects.*
111922d9 0549 464b 8b1f d8240a5f7900 10

NDC 68180-772-06 is a prescription drug with Amlodipine, Valsartan, and Hydrochlorothiazide. It is available in film-coated tablets and is manufactured by Lupin Limited of Goa 403 722 in India for Lupin Pharmaceuticals in Baltimore, Maryland. Each tablet contains 10mg Amlodipine besylate USP equivalent to Amlodipine, 160mg Valsartan USP, and 12.5mg Hydrochlorothiazide USP. The complete dosing instructions can be found in the accompanying prescribing information. It should be stored at 25°C (77°F) with excursions permitted to 15°t0 30°C (59° to 86°F) and protected from moisture.*
111922d9 0549 464b 8b1f d8240a5f7900 11

This is a description of a prescription drug. The drug has the NDC number 68180-773-06 and is manufactured by Lupin Pharmaceuticals, Inc. It contains Amlodipine besylate USP equivalent to 5mg and Valsartan USP 160mg and Hydrochlorothiazide USP 25mg. Each tablet is film-coated and the package contains 30 tablets. It is recommended to store the drug between 15°C to 30°C (59°F to 86°F) and protect it from moisture. The accompanying prescribing information should be consulted for complete dosing instructions.*
111922d9 0549 464b 8b1f d8240a5f7900 12
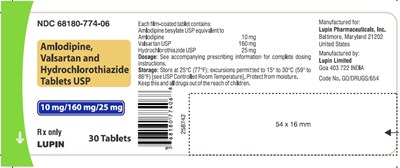
This is a prescription drug called Amlodipine, Valsartan, and Hydrochlorothiazide Tablets USP manufactured by Lupin Limited in Goa, India. Each tablet contains Amlodipine besylate, Valsartan, and Hydrochlorothiazide, with a dosage of 10mg, 160mg, and 25mg, respectively. The enclosed prescribing information provides detailed dosing and administration instructions. The drug should be stored at room temperature between 15°C to 30°C, and protected from moisture. This drug should be kept away from the reach of children.*
111922d9 0549 464b 8b1f d8240a5f7900 13

This is a prescription drug with the NDC code 68180-775-06. It is a film-coated tablet that contains Amlodipine besylate USP equivalent to Amlodipine 10mg, Valsartan USP 320mg, and Hydrochlorothiazide USP 25mg. The manufacturer of this drug is Lupin Pharmaceuticals, Inc. The dosage information can be found in the accompanying prescribing information. The tablets should be stored at room temperature and protected from moisture. It is important to keep this drug out of the reach of children.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.